The latest analysis done by scientists from the National Institute of Standards and Technology (NIST) and the Massachusetts Institute of Technology (MIT) takes us one step closer to making crystals a battery ingredient candidate. The scientists’ efforts resulted in a clear picture of the molecular structure of manganese-based crystals.
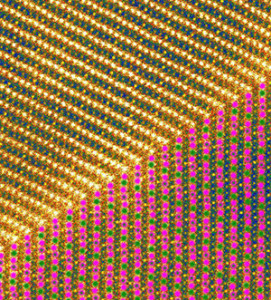
The top of this image shows sodium filling layers of crystal (as represented by 1 bright yellow and three darker ones). The bottom shows the layers’ magnetic ordering as depicted by the green and purple dots. Each color represents the two different charge states of magnesium. The green in purple dots represent the mixture of the two different charge states.
This picture will allow scientists to explore the magnetic and electronic properties of crystals in general. Crystals are known to possess qualities that have potential for use in batteries. They can all store sodium, an atom that can flow in and out of the layers of the crystal every timeelectricity is applied. This means they can store chemical energy – which is an important feature needed in rechargeable batteries.
Not only can crystals store energy, some also have capabilities to be low-temperature superconductors.
Despite these qualities, however, they are not popular as a battery candidate because their molecular structure is so convoluted that it is difficult to pinpoint what they can do. This makes it difficult for manufacturers to improve their performance.
The current analysis was made possible because of electron microscopy and neutron beams, which allowed the scientists to play with the interactions between the individual atoms of manganese-based crystals. It was revealed that in crystals, sodium is absorbed in a way that is unlike any other. The unusual pattern signifies that the atoms in the different parts of manganese-based crystals have varying charges and magnetic moments.
While all of this does not mean it will be used as a battery immediately, scientists can nevertheless understand what goes on inside the molecular structure of these crystals. There is now a basis to tailor the properties of crystals by changing the transition metals or sodium content to make it a more probable battery candidate.
Related Articles:
Is Electricity a Type of Energy?
A Sodium Ion Battery Could Replace a Lithium Ion Battery in the Future



