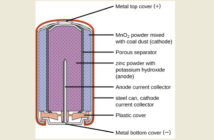
There are many different types, shapes and sizes of batteries on the market. These range from the heavy duty lead-acid ones we make and sell, to the tiniest ones in hearing aids. These all do their work with the same chemistry. This article is a brief introduction to how batteries work.
The Three Main Contributors to How Batteries Work

The three main parts of any battery are two (usually) metal electrodes of different materials, and an electrolyte. The electrolyte separates these electrodes while allowing energy to flow between them.
However this flow only occurs when there is a corresponding flow of electricity between the electrodes and an external device. That’s all there is to the basics of how batteries work although we have a bit more science to tell. We call the negative electrode the cathode, and the positive one the anode. The energy itself is in electrons and ions that shuttle between the two.
How the Electrons and Ions Shuttle Between the Two Electrodes
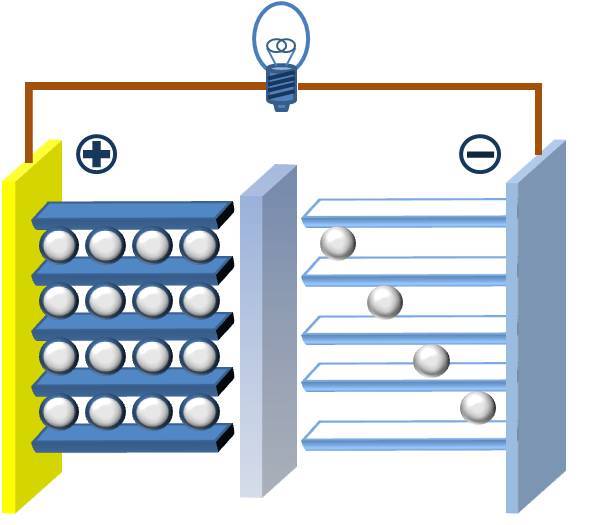
Let’s assume the battery is in a charged state. When we connect it to an external device, the chemical in the anode sends electrons to the negative terminal and ions pass through the electrolyte. Meanwhile, the cathode at the positive terminal receives these electrons, thereby completing their flow.
This is how all batteries work regardless of their type, shape and size. The ions take energy through the electrolyte. While the electrons flow as electricity in the external circuit thereby generating an electric current. Some batteries are disposable. When they are flat they are only good for recycling their materials.
However an increasing number of batteries can recharge by reversing the flow of ions through the electrolyte. The battery industry also calls this process ‘recycling’. Now you know how batteries work we hope you enjoy the rest of our posts.
Related
Safe Lithium Battery with Graphite Cathode
New Lithium Electrolyte Extends Temperature
Preview Image: Side-By-Side Battery Pack