Researchers at Rice University in Houston, Texas seemed well pleased with themselves on December 31, 2019. They told Design News they had found a way to tune phosphate-based lithium-ion cathodes so they worked better. Their simulations revealed placing ‘specified defects’ in them bolstered performance. However, on January 14, 2020 they admitted you can push lithium ions too far.
This Might Have to Lead to Charging Two Times Faster
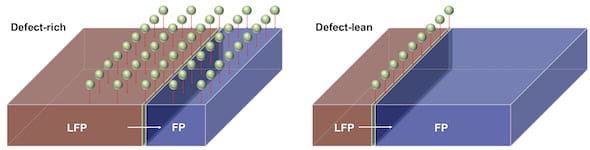
The researchers focused their attention on a critical point during lithium battery cycling. That is when the cathode morphs from an iron phosphate to lithium iron phosphate as the lithium ions shuttle.
The researchers ignored convention by adding ‘antisites’ defects to their cathode crystal lattices. Battery researchers customarily ignore these antisites because they believe they restrict lithium ions moving inside the lattice. “We think we can turn defects into friends, not enemies, for better energy storage,” graduate student Kaiqi Tang said. That was because his antisite ‘defects’ provided a wider surface for reactions and hence lithium migration.
Then He Agreed You Can Push Lithium Ions Too Far
Two weeks later the Rice researchers took a double somersault. Materials scientist Ming Tang told Science Daily the latest x-rays showed the defect-laden cathodes could fracture. This was especially the case during rapid charging and discharging. You can push lithium ions too far he admitted. So what went wrong?
Lithium normally moves onto the cathode surface, where after it spreads smoothly to the center. There’s usually a flat boundary between lithium-poor, and lithium-rich regions. However, the second round of x-rays showed a ‘finger like’ boundary, for example as when injecting water into oil. This altered the boundary shape, increased the stress level and could cause the cathode to crack.
However, Kaiqi Tang is not disillusioned. He believes there is a ‘sweet spot’ for the number of antisites. There must be sufficient to enhance performance, but not enough to cause instability, he says.
Related
How Batteries Work – A Brief Introduction
Electrochemical Reactions in Batteries
Preview Image: Kaiqi Yang and Ming Tang
Video Share Link: https://youtu.be/rFOPCA_r70k




