Lead battery sales bounced back in 2021, after COVID increased the need for reliable alternate energy at hospitals. A number of automotive starter batteries may also have needed replacing as companies reopened for business. Lead acid battery plates may be one of the most reliable and cost-effective technologies around, but they do require recharging.
Will the Threat of Extreme Weather Accelerate Demand?
It seems we are going to pay the penalty for ignoring global warming as a nation. We daily read reports of upticks in floods, hurricanes, and raging wild fires. Households need back up power for charging phones when the grid goes down, and nowadays occasionally respirators too.
But most consumers still regard their lead batteries as ‘black boxes’. We dedicate this post to explaining lead acid battery plates inside them. Because these are key to quick and easy grid failure solutions at home. Solutions we can rely on time and time again.
How Lead Acid Battery Plates Do Their Work
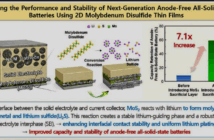
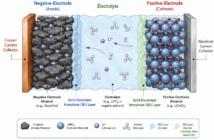
The plates in lead batteries are made of lead, as the name suggests. This is soft enough to take on, and release ions during discharging and recharging. Early designs added metalloid antimony to harden the soft metal and improve its performance. However, this did eventually cause corrosion at the positive sides of batteries
Nowadays battery engineers tweak this compound further to improve electrical conductivity. Selenium mineral is popular as it refines the lead and puts paid to positive grid corrosion. This also extends the operational life of deep cycle lead batteries considerably.
There have been many more improvements to lead acid battery technology during the century-and-a-half since its inception. However, remarkably the basic structure is still the same. We wonder how many other technologies could claim this record, as reliably as our lead batteries do!

Recent Posts
Further Advantages of Batteries in Freight Cars
Battery Electric Freight Could Save 50% Energy
Preview Image: Battery Harvesting Solar Energy