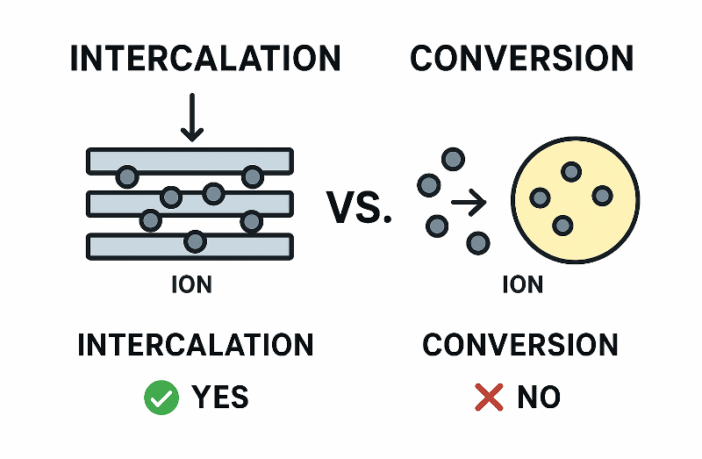
Intercalation is a process whereby qualifying ions insert themselves between layers of material structures, without fracturing them. This is the basis for lithium-ion batteries’ superior performance. However, many battery types do not intercalate, as they store and release energy. Sodium-ion and potassium-ion batteries are the only other exceptions that spring to mind.
More About -Ion Battery Types That DO Intercalate
- Lithium-ion battery ions leave the cathode during charging, and intercalate into the graphite anode.
- Those same ions de-intercalate from the anode during discharging, and return to the cathode.
- Sodium-ion and potassium-ion batteries work on a similar principle, but their materials are different.
- Magnesium-ion and zinc-ion batteries follow the same pattern, although they are still largely experimental.
Popular Batteries That Use Different Chemistries
Many rechargeable battery types do not intercalate, as we describe above. They use a process called a conversion reaction instead as they discharge. We can compare them as follows:
- INTERCALATION – Ions insert into a crystal structure without breaking it apart.
- CONVERSION – Ions react with electrode material to form new compounds.
- Both of these processes can be reversed, by recharging either battery type.
- LEAD-ACID – Lead-Acid Batteries use a conversion reaction to form lead sulfate.
- LITHIUM – Lithium-sulfur and lithium-air batteries also form new compounds.
- NICKEL – Nickel batteries use conversion and alloying reactions, not intercalation.
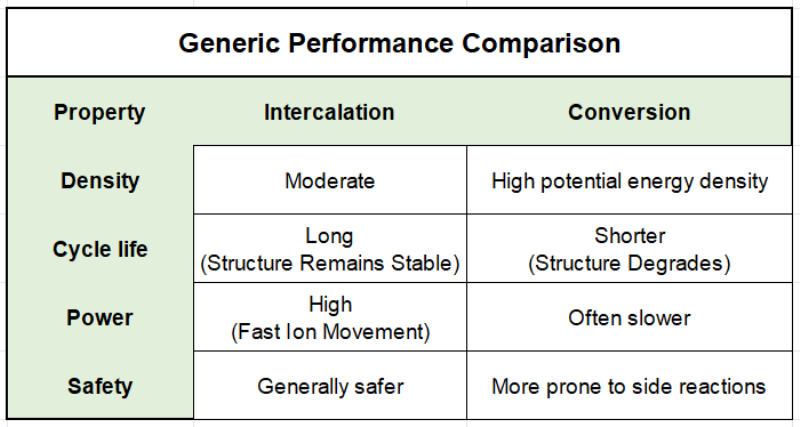
In summary then, Intercalation batteries are generically stable, longer-lived, and dominate current technology. Whereas conversion batteries can, in theory at least, store more energy but tend to degrade faster.
More Information