Zinc-iodine batteries are naturally safe. Their low cost, and low environmental impact make them great candidates for large-scale battery energy storage. Unfortunately though, engineers have to date been unable to build stable iodine cathodes, lending themselves to mass production. That said, a novel University of Adelaide zinc-iodine battery design appears to have broken through.
A University of Adelaide announcement confirms that their researchers bypassed traditional wet-mixing iodine methods. Iodine is a mineral essential for making thyroid hormones in our bodies, which regulate metabolism and other bodily functions.
Adelaide Accepts the Challenge of Mass-Producing the Batteries
Zinc-iodine batteries have long been promising candidates for large scale energy storage projects. That’s because they out-perform lithium-ion in terms of resource availability, and thermal stability controlling overheating.
The batteries also have fast redox reactions, and there is, moreover, an abundance of iodine in sea water. However, the catch has persistently been the lack of a scaleable way to produce iodine cathodes. This was what the Adelaide zinc-iodine battery team set out to resolve, and they did it!
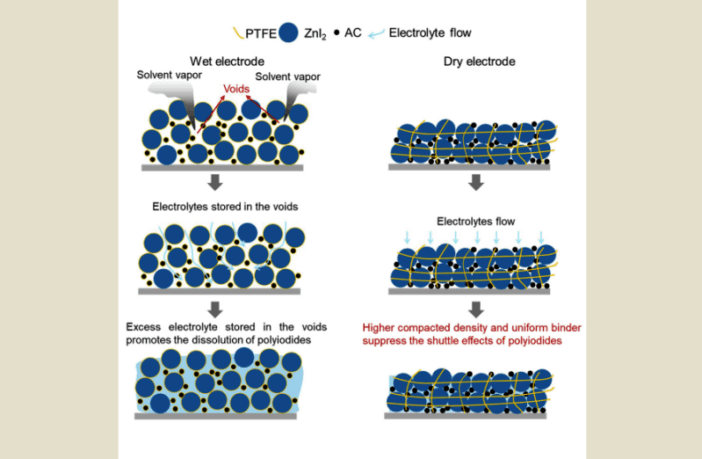
A New Electrode for Aqueous Zinc-Iodine Batteries
“We mixed the active materials as dry powders, and rolled them into thick, self-supporting electrodes,” the director of the center for materials in energy and catalysis who led the team, explains.
“At the same time, we added a small amount of a simple chemical, called 1,3,5-trioxane to the electrolyte, which turns into a flexible protective film on the zinc surface during charging.
“This film keeps zinc from forming sharp dendrites. These are needle-like structures that can form on the surface of the zinc anode, during charging and discharging. This can short the battery if not controlled.”
The higher-capacity zinc-iodine pouch cells the team built, retained 88.6% of their capacity after 750 cycles. The button cells achieved a higher 99.8% after 500 recharge-discharge cycles. Industries requiring large stable energy banks, could consider adopting this technology to their advantage.
More Information
Novel Current Collector for Zinc-Ion Batteries
Zinc-Based Batteries Do Have a Future
Preview Image: Much More Energy Per Battery