Did you ever wonder whether an alkaline battery lying on your workbench is still fresh? If it is not, then it might be about to burst and cause an alkaline battery leak. These things matter. If it is resting on a printed circuit board, you could be up for an expensive replacement. If you lent your multi meter to your best mate for the weekend what happens next?
You Try the Bouncy Alkaline Battery Test

If you drop an alkaline battery on either end approximately vertically, then it will bounce. Most things do thanks to gravity and Newton’s law of opposite and equal reactions.
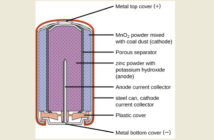
Interestingly enough a bunch of students at Princeton University found a fresh battery bounces less than a discharged one. They ascribed this to the zinc electrode changing to zinc oxide as the battery goes progressively flatter.
“As you get more and more zinc oxide, and the zinc oxide begins to appear everywhere in the zinc layer, the battery gets bouncier and bouncier,” the lead researcher explains.
A Few More Things to Know Before Bouncing Batteries
Two things are relevant here. First, zinc is a common component of many golf balls where it adds to bounce. Then secondly, the process is not a straight-line one. Alkaline battery-bounce levels out when the battery approaches half its run time. The Romeo Club for Retired Men Eating Out in Cincinnati investigated further and released this video:
We find their analogy of an anti-bounce hammer intriguing. As the squishy alkaline battery electrolyte dries out, it changes from being a ‘shock absorber’, to an ‘accelerator’. We would like to add one thought to the debate. Before you bounce an alkaline battery, please make sure the case is intact and not swelling even slightly. Because it might otherwise leak. Provided you wear safety glasses, you should have covered all your bases.
Related
How to Clean Up Alkaline Battery Leaks