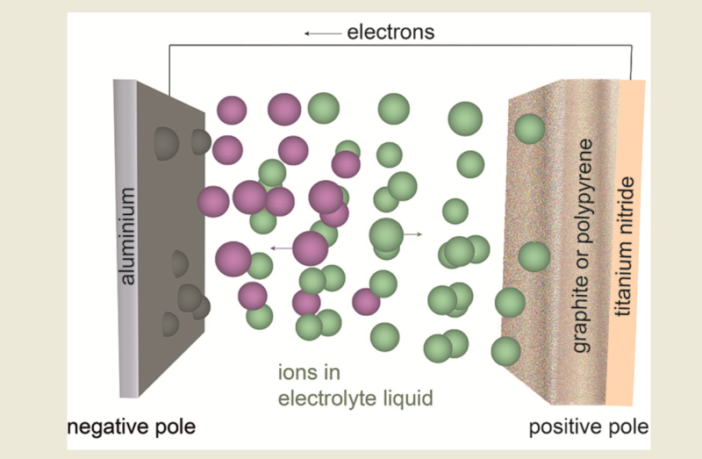
A battery containing sufficient electrical charge, is capable of transmitting an electric current through an external circuit. It does so by releasing some of its store of electrons from its cathode electrode. This in turn disturbs the balance between its battery fundamentals, which it addresses by triggering a flow of ions to its anode electrode.
Battery Conditions Essential for These Fundamentals
A battery requires the following conditions to be present before it can do the above work:
- Sufficient electrons remaining on its cathode from its charge or recharge.
- Sufficient electrolyte to allow the compensating ions to shuttle to the anode.
- A conductive link between the two terminals matching the electron output.
The electrolyte must comprise a solvent containing the ions we referred to above, plus one or more chemicals that trigger the process. This electrolyte may be water, an organic liquid, or even a solid substance. However, its composition must match the requirements of the two electrodes.
There Is More Than Just That To Battery Basics
Battery fundamentals extend to more than just matching the physical components. That’s because the salt ions in the electrolyte must be able to move fast enough to meet the demands of the application. Battery performance therefore depends equally on the composition of the internal chemicals too.
The temperature inside a battery also influences its performance. This may depend on chemical reactions inside the battery, or the ambient temperature outside. If the temperature falls too far, and freezes the electrolyte, then too few ions may be able to shuttle to produce a useful current.
Extreme high and low temperatures may cause battery components to expand or contract, resulting in personal injury or damage. This may at the very least result in diminished performance, even after battery temperature returns to normal. Artificially warming a battery to increase the rate of discharge may evaporate the electrolyte, leading to early battery failure.
More Information
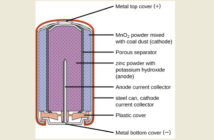
Different Battery Types & What Happens Inside