Solid-state lithium metal batteries with solid electrolytes, are attracting worldwide interest. This is because the world needs something to replace lithium-ion technology, which is approaching its topping out point. Researchers at Tohoku University in Japan have been examining the options for creating solid electrolytes for batteries, and have discovered something most interesting.
Using Garnet-Type Oxides in Solid Electrolytes
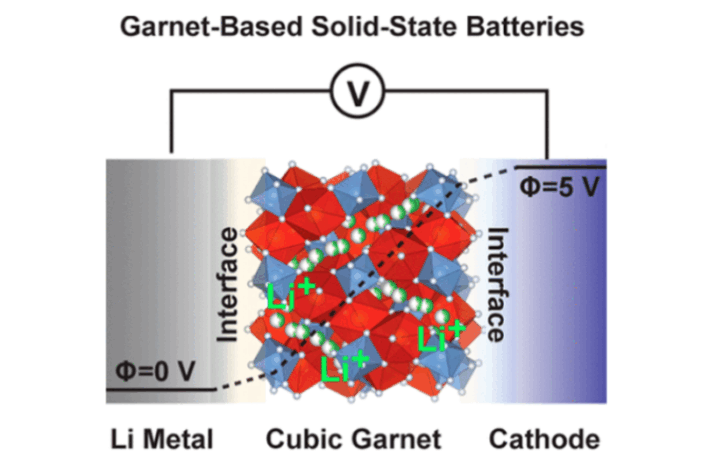
Garnet-type oxides are an attractive proposition for solid-state lithium metal batteries. This is because their chemical structure allows high lithium-ion conductivity, while maintaining good chemical stability against lithium metal anodes.
Garnet-type oxide Lithium La₃Zr₂O₁₂ (LLZO) is high up on the list of possibilities. However, there are challenges to creating thin, dense, and defect-free ceramic membranes from it, and this prevents commercial scale applications.
The current manufacturing method involves sintering at 1000 °C / 1800 °F for several hours. This manufacturing process binds powdery materials by heating them, to a high temperature below their melting point. However, this method consumes energy that global warming calls on us to use sparingly.
Pressure-Assisted Sintering for Solid Electrolytes
Clearly, we need a lower-energy solution for creating solid electrolytes for batteries using garnet-type oxides. This is especially true, because high temperature sintering leads to lithium evaporation, while costing more and discouraging commercialization.
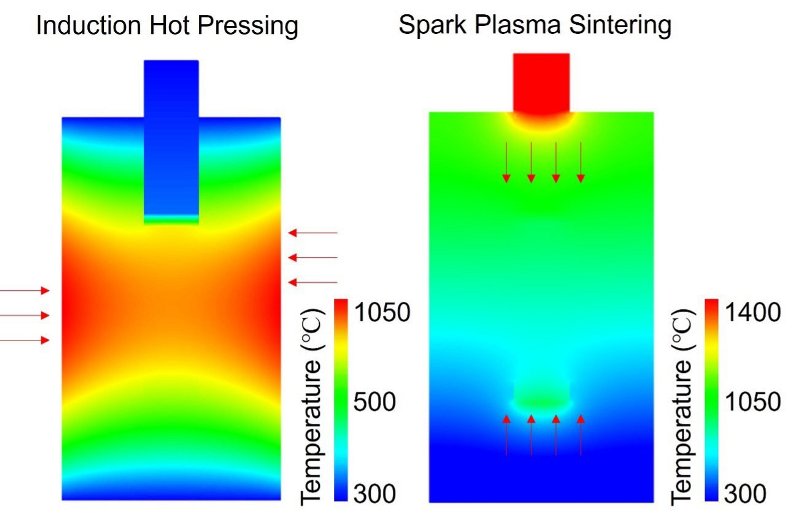
The team at Tohoku University were already aware of research into hot pressing, and spark plasma sintering, as alternative approaches with the latter preferred. However, their research which we report on here presents a different picture:
- Both methods achieve nearly full densification in under five minutes.
- There are no significant differences in ionic conductivity or micro-structure.
This finding reveals that applied pressure and heat drive densification just as in hot pressing. There is therefore no special plasma effect. The decision criteria are cost, equipment, and scalability factors.

More Information
Solid State Battery With Multilayered Electrolyte
Thin-Film Coatings on Solid State Batteries
Preview Image: Garnet-Based Solid-State Batteries