We have been writing about fuel cells lately, which relate to batteries but are quite different. A kid wrote in and said “that’s all good stuff, but I was hoping to learn how a lead battery makes electricity”. The editor said “get to it right away”. So here’s our explanation of what has been happening inside our lead-acid batteries for a very long time.
How We Make Electricity With Our Lead Batteries
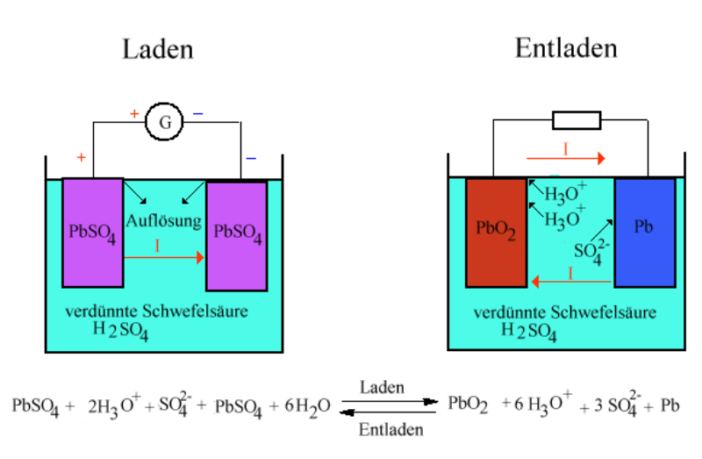
We chose our feature image even though it is in German, because it explains the principles so perfectly:
- The battery on the left is charging, while the one on the right is discharging.
- The positive and negative electrodes are both immersed in the electrolyte.
- Notice how their chemical composition is different depending on their state.
The negative electrode is spongy, or porous lead (Pb), while the positive one is lead oxide (PbO). They rest in a solution of dilute sulfuric acid (H2SO4) mixed with water, in a ratio of 1:4. A chemically permeable membrane prevents them touching and short-circuiting.
Discharging and Charging Our Batteries
A lead battery makes electricity when we discharge it, by sending electrons through an external circuit. The secret sauce behind this is the chemistry inside:
- Lead sulfate crystals (PbSO4) form on both electrodes during a discharge.
- These come from the sulfuric acid, effectively diluting that solution further.
- The voltage of the battery gradually reduces as the crystals accumulate.
- The electrodes in a discharged battery become completely covered over.
- As they have now become the same material, the chemical process pauses.
We can replenish the battery at this stage, by recharging it with electricity. This returns the electrodes and electrolyte to their near-original state. However, if we leave the battery in a discharged state for a long time, larger lead sulfate crystals may form, and permanently limit the battery’s capacity.
More Information
Depth of Discharge in Storage Batteries