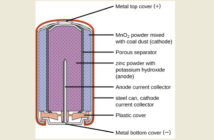
We would find it impossible to describe how batteries work without mentioning ions, because these are the basis for stored electricity. Connecting a charged battery to a device produces a stream of positive ions, and negative electrons. Ions in batteries shuttle between electrodes through an electrolyte. While electrons pass through the device itself, causing it to operate.
Where the Ions in Batteries Come From

All ions in batteries are functionally similar, although their chemical composition varies between battery types. We define ions as particles having a non-zero electrical charge, which is thus either negative or positive.
Cations have positive charges, while anions have negative ones. This is the basis for calling the positive electrode a cathode, and the negative one an anode. Ions from pairs of chemicals may have a tendency to attract each other, and form ionic compounds for example sodium chloride.
Why We Call Ions ‘Ions”

Michael Faraday, the English chemist and physicist coined the term ‘ion’ in 1834. Through observation, he knew that certain metals dissolved at one electrode. But then formed a new one at the other electrode, after passing through a solution containing water.
Faraday suspected an unknown ‘species’ moved through the solution like a conveyor belt. So, he named the new species ‘ion’ after the Greek word ‘ἰόν’ denoting going, and to go. He also brainstormed the words cation for a positively charged ion, and anion for a negatively charged one.
A Universe Filled With Ions
The ions in batteries are particles and thus minutely solid. We call ions in a gaseous state plasma, and plasma may be 99% of all visible matter in the Universe. We find it amazing to think that the sun, the stars, and the space between the planets share a common denominator with our lead-acid batteries. Ions: Those little power houses without which life could not be the same.
Related
Electrical Conductors We Call Electrodes
Role Plasma Plays in Nuclear Fusion
Preview Image: Alkaline Battery Disassembled