The key to increasing lithium-ion battery energy-density is packing more lithium ions in the cathode. Engineers layer alternate lithium and transition materials such as manganese or sulfide to achieve this. Increasing the amount of lithium however reduces the transitional materials providing electrons to the external circuit. French scientists have found a way to improve the performance of lithium-rich layered sulfides.
New Way to Boost Lithium-Rich Layered Sulfides

Anion redox reactions are a promising new way to increase rechargeable capacities in alkaline-ion batteries. Scientists at College de France and Sorbonne University are seeing ways to produce a commercial application.
That’s because lithium-ion is the best solution we currently have for electronic devices. However, its density has hit a chemical ceiling because lithium rich cathodes are less reversible. Moreover, they produce significantly lower voltage after their first charge. Phys Org reports the scientists applied their minds to resolving cathode disintegration during cycling, as this causes an early, significant fall in battery density.
New Cathode Ribbon Structure Shows Promise
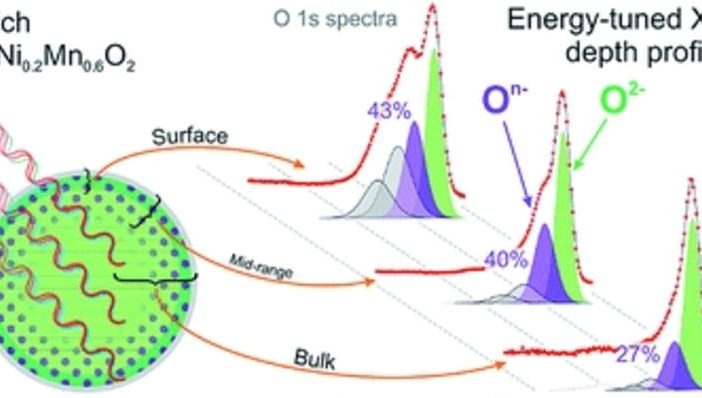
Collaborating scientists at University of Oxford, Robert House used spectrometers to study two closely-related cathode materials. They noticed oxidized gas molecules forming in the middle of a cathode. This disturbed the transitional metal layer, resulting in first-cycle voltage loss.

The first test sample used a honeycomb form to store excess lithium. However, the second one that performed better had a ribbon structure. This different configuration of lithium and transition metals suppressed the formation of oxygen gas. These are early days, and the ribbon structure is not entirely stable yet. Despite this, the study does help explain how lithium-rich layered sulfides behave, and offers a possible pathway to better, denser batteries.
Battery research is a slow, often disappointing process that seldom produces commercial solutions. None the less, each stage adds its own value and helps create the road to progress. Thus, we welcome each new initiative and share it on these pages.
Related
Electra-Chemical Reactions in Batteries
Trapping Polysulfide Molecules through Nanotechnology
Preview Image: Depth-Dependent Oxygen Redox Activity