Electrochemistry concerns itself with the relationship between electrochemical difference, and identifiable chemical change. An electrochemical cell is therefore a battery that generates electrical energy from chemical reactions. Electrochemistry in lead-acid batteries is a special case of this, because the materials are unique to this particular product.
Electrochemisty and Materials in Lead-Acid Batteries
| A discharging lead-acid battery comprises pairs of lead sulfate plates, separated by dilute sulfuric acid. This acid is less-concentrated than ideal, because some of the sulfur has migrated as crystals to the two plates.
After the battery has fully discharged, both plates are chemically identical, and there is thus no difference in their chemical potential. The device can no longer deliver the energy we need. However, this composition changes when we recharge the battery:
|
Materials in Modern Lead-Acid Batteries
We could demonstrate the electrochemistry in lead-acid batteries by using two plain lead plates. However, the amount of energy would be minimal, and transient too, due to the size of the surface area. Gaston Planté originally solved this challenge with longer sheets in a jelly roll configuration.

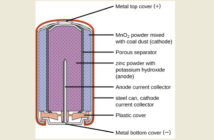
But Camille Faure devised the more elegant design in our first image in 1880. He coated a lead grid – with a larger effective surface area – with a paste of lead oxide, sulfuric acid, and water. Then he cured this paste during the battery’s first initial charge, converting its plates to electrochemically active material.
This method has continued, with minor modifications for over a century, although the experimental work continues unabated. Lead-acid batteries have been an industry stalwart for a long time, among other things because they are solid, reliable, and cannot overheat to the extent they catch fire.
More Information
Lead-Acid Battery Energy Storage