The lavender plant is native to many countries with moderate, mediterranean-style climates. The flower has been popular for centuries, on account of its naturally occurring linalool oil that our ancestors used for bathing. Scientists from Max Planck Institute of Colloids and Interfaces have discovered that linalool extends sodium-sulfur (NaS) battery lifespan. Sodium sulfur flower power, what’s next?
How Polysulfide Shuttling Degrades NaS Batteries
A molten-salt-type sodium sulfur battery (NaS) uses liquid sodium and liquid sulfur electrodes. In principle, this combination achieves similar density to lithium-ion while using inexpensive, non-toxic materials. However, several characteristics prevent wide use of sodium sulfur chemistry:
- The combination requires a high operating temperature between 300 and 350 °C.
- Sodium and sodium polysulfides are highly corrosive preventing their use in vehicles.
But perhaps the greatest impediment to sodium sulfur battery roll-out is a phenomenon called polysulfide shuttling. This anomaly occurs as sodium polysulfides dissolve in the electrolyte. This event leads to severe anode corrosion, low coulombic efficiency, and rapid loss of capacity.
Polysulfide shuttling hence stands in the way of sodium sulfide molten batteries competing with lithium-ion for bulk energy storage. And so this option remained in the background, until the advent of the sodium sulfur flower power experiment.
Flower Power Prevents Sodium Sulfur Polysulfide Shuttling
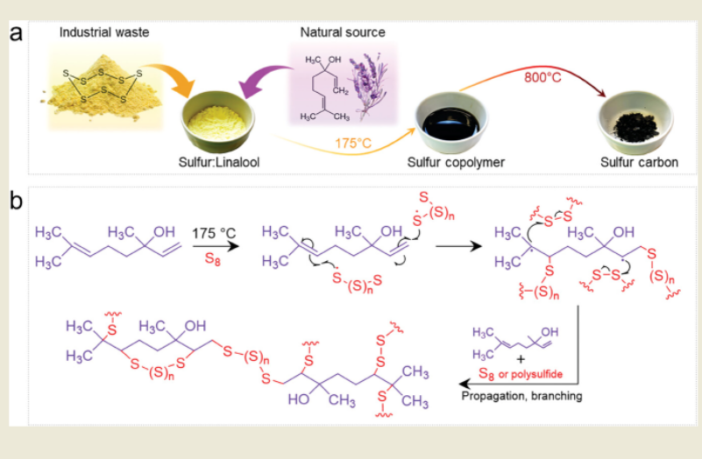
The team at Max Planck Institute of Colloids and Interfaces combined lavender oil (linalool) with sulfur. This created a unique material that prevented persistent polysulfide shuttling.
They describe this novel combination as ‘acting like a confinement cage’ for the sulfur and polysulfides. The nano pores in the lavender-sulfur compound were small enough to trap the polysulfides, but large enough for the smaller sodium ions to pass through.
The sodium sulfur flower power solution proved stable, and stood the test of time. The Max Planck Institute press release confirms the ‘lavender-tweaked battery’ retained more than 80% of its capacity after 1,500 lab tests, over three months.
“If we look at nature with a creative eye, it offers solutions to many challenges of the energy transition,” the team leader observed. “I am confident that our results will soon find their way out of the lab into real-world applications.”
More Information
A Sodium Ion Battery Could Replace a Lithium Ion Battery in the Future
Molten Salt Batteries in a Class of Their Own
Preview Image: Synthesis and Inverse Reaction