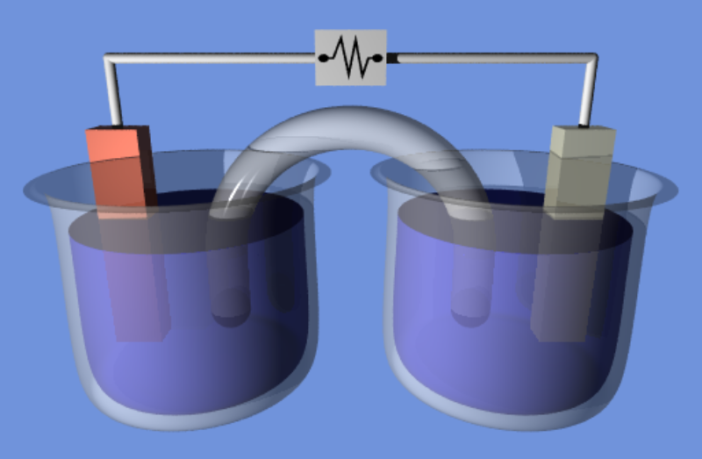
Our first two posts in this short series spoke of the electrodes in a battery, and how they compete for electrons between them. You’ll find two links to those posts below. But today we turn our attention to the secret battery manager electrolyte. For this is what enables the electrodes to work together and produce the electricity.
The Role of the Silent Battery Manager Electrolyte
The electrons have a negative charge. We need something else to balance them out, because the natural order tends towards neutral equilibrium. The electrolyte facilitates this, by allowing balancing positive ions to flow through it. This material can be a liquid, a gel, or a solid substance.
- A chemical reaction at the anode produces negative electrons that flow through an external circuit to the cathode.
- A matching amount of positively charged ions simultaneously flow into the electrolyte from the anode.
- The cathode in the meanwhile attracts those positive ions to restore its own electrochemical balance.
- However, in some cases the cathode releases negatively charged ions into the electrolyte instead.
The Electrolyte is the Electrochemical Umpire
The secret battery manager electrolyte hence plays an essential balancing role in keeping a battery cell’s house in order. To summarize at this point in the series:
- The external circuit from the anode allows negative electrons to flow through, and energize a connected device.
- The internal circuit through the electrolyte to the cathode balances the internal system keeping the reaction going.
Hence, the electrolyte is as important as the external device running off the battery. Our next article will explain how to recycle the critical ions back to the anode, so we can use the battery again.
Breaking News
How the Chemistry in a Battery Works
Standard Potentials for Half Reactions
Preview Image: Diagram of An Electrochemical Cell
Detailed Explanation by Australian Academy of Science