Electric currents can also be found in organic materials. Who would have thought that an orange you have picked up from the grocery can actually power up a small light bulb? This popular experiment is created to open our eyes to the many possibilities of producing electrical phenomena using nature’s very own crops.
What You’ll Need:
- Citrus fruit (lime, lemon, orange, grapefruit, etc.)
- A piece of copper (nail, screw or wire), 2″ long
- A piece of zinc (nail, screw or galvanized strip), 2″ long
- Holiday LED light, or any 2-inch lead bulb containing a wire which can be connected to the nails
- 1 cable with alligator clips
- A voltmeter, for measuring the electric current (optional)
Steps in Making a Fruit Battery:
- Roll the fruit firmly with your palms or at the top of the table. This is done to break the small sacs to allow its juice to flow inside the fruit. Be careful not to break the skin.
- Insert the copper and zinc strips into the center of the fruit, but keep them 2″ apart from each other. Do not puncture through the other end of your fruit battery.
- Optionally, you can measure the electrical current by clipping one end of the voltmeter on the copper strip, and the other end on the zinc strip. Remove the voltmeter after checking the amount of energy.
- Remove about 1″ of the insulation from the leads of the bulb. Wrap one bulb wire around the zinc strip and another on the copper strip. Use the cable with alligator clips to keep the wires in place.
- As you connect the wire onto the second metal strip, your bulb will light up.
How does it Work?
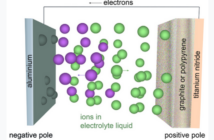
The atoms in Copper (Cu) attract more electrons than the atoms in Zinc (Zn). Copper serves as a positive electrode and Zinc serves as a negative electrode. Zinc represents the negative pole because of its ability to donate its own electrons to another metal. Copper represents the positive pole because it is less reactive than Zinc, and only attracts electrons.
In order to produce electricity, the Copper and Zinc strips must be covered in a conductive solution. This makes citrus fruits perfect for the job. The juice inside these fruits has acids and enough pH level to conduct electricity.
With the citrus fruit’s help, the reactions between the electrodes and the solution charges the light circuit with a continuous electrical stream.
The energy does not come from the citrus fruit itself. Electricity occurs because of the chemical change in Zinc, which is oxidized inside the fruit. The Zinc’s exchange of electrons with the Copper releases energy which is turned into electricity.
Discover more possibilities of creating batteries out of a potato or a cola!
Related articles:
Image Credits: Caleb Charland via This Is Colossal