Researchers from Sandia National Laboratories created the smallest battery in the world from a lithium-based rechargeable battery that can be used to run minuscule electronic devices. The battery is a cross between a supercapacitor and a battery.
Super capacitors can deliver more power than batteries. The nano battery is six times finer than a bacterium. Its thickness is less than the wavelength of visible light, meaning not as much as the lowest wavelength which is 400 nm. It is about 150 nanometers wide, one seven-thousandth finer than a human hair which is 50 microns, and over 60,000 times smaller than an AAA battery. It consists of a three millimeters bulk lithium cobalt cathode, an ionic liquid electrolyte, single tin oxide (Sn02) nanowire 10 nanometers long, and 100 nanometers in diameter.
Lithium cobalt oxide (LiCoO2) is a chemical compound used in lithium-ion batteries positive electrodes. Single tin oxide nanowire anode is a stable and high-capacity material used for Li-ion batteries.

Each battery is a nanowire with half of the wire working as a positive electrode and the other half as a negative electrode. Arrays of these nano batteries could power biological and chemical sensors, implantable medical devices, and microscopic wireless networks. They could also be embedded in everyday objects and computers.
To create this battery, scientists coat the nanowire template with copper and fill the pores halfway with nickel alloy to create anodes. A thin layer of polyethylene-oxide gel was added to act as an electrolyte and insulator from other nanowires. The remainder of the pore was filled with polyaniline to create the cathodes, and, to complete the circuit; a layer of aluminum was placed on top.
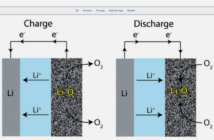
The nanowire based materials in Lithium-ion batteries has offered more significant improvement in energy and power. To understand the fundamental mechanism by which the batteries work scientists formed the battery inside a TEM (transmission electron microscope) so that researchers can see the charging and discharging of the battery in real-time.
Researchers found that the tin nanowire rod doubles in length during the charging of the tin oxide nanowire instead of the common belief that batteries swell across their diameter. This can avoid short circuits that shorten the battery life and prove that nanowires can sustain large stress without breaking, which makes them better candidates than electrodes.
Nanobatteries are not without limitations because after being charged and discharged 20 times, they lose their ability to hold a full charge and researchers are working to address these limitations by trying a different kind of electrodes and polymer thickness.
Related articles: