We established in a previous post how electrons travel between electrodes in a battery to deliver energy. And that we can reconstitute the spent energy in a secondary, rechargeable cell, by reversing the electron flow. Now it’s time to share how this electrode chemistry works, as it is at the heart of the entire process.
How Electrode Chemistry Works in Principle
Two electrode chemical reactions occur simultaneously, so we need to get a grip on them:
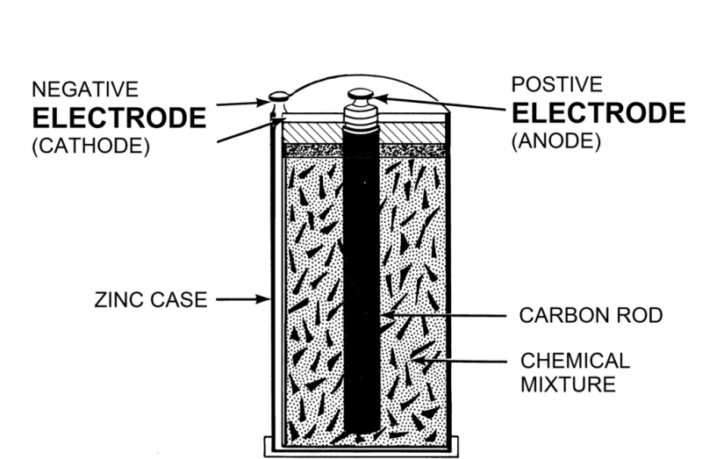
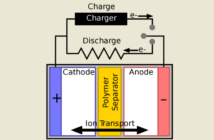
- The negative anode electrode interacts with the electrolyte to generate electrons.
- These electrons wait while the positive cathode electrode prepares to receive them.
Science calls the process whereby anodes exchange these electrons a reduction-oxidation reaction, shortened to redox. We’ll understand electrode chemistry better if we analyze the stages:
- One half reaction occurs at the anode, and the other half at the cathode.
- Science calls the loss of electrons at the negative anode, oxidation.
- While it calls the gain of electrons at the positive cathode, reduction.
You may find the use of these two terms unusual. However, they are the origin of the expression ‘reduction-oxidation reaction’, or redox for short.
Each of these half-reactions has a standard potential, or ability to provide or receive electrons. Any two materials with different standard potentials can form a battery electrochemical cell.
The Science of Pairing the Right Electrodes
Battery design is the science, or might we say the art of pairing an anode with greater negative standard potential than the corresponding cathode. This provides an easier pathway for the movement of the electrons, so we can use them to power our devices.
Current battery research is constantly on the hunt to develop high-powered electrodes that don’t have an electronic ‘tug of war’. Lithium-ion technology has taken us a long way down the journey. Although there have been occasional spectacular results. After electrode dendrites reached through the electrolyte, and short-circuited the battery.
More Information
How to Make a Battery Cell at Home