Solid electrolytes hold promise of safe, high-energy storage systems. That’s because, in theory at least, they should deliver stable electrode interfaces allowing fast ion movement. We present a novel idea for a solid electrolyte as fast as liquid that achieved this goal in a laboratory.
This contribution comes from a team of Chinese scientists at Shenzhen Institutes of Advanced Technology. They tackled a fundamental issue. This was to simultaneously achieve fast ion transfer in all-solid-state lithium batteries, together with mechanical electrode compatibility.
The Problem with Solid Electrolyte
The inorganic electrolytes in all-solid-state batteries, do a great job of conducting lithium ions in rigid crystal lattices. But they perform badly when it comes to creating a consistent interface with the electrodes.
The preferred solution has been maintaining high pressure between these two elements. But unfortunately, this restricts the amount of energy that the battery can store. It is also difficult to maintain the pressure in commercial applications.
This really is a double bind. That’s because the alternative, polymer electrolytes achieve close electrode contact, but are not good conductors of lithium ions. Composite electrolytes blending both materials have not been successful to date either.
A Problem Shared As Fast As Liquid
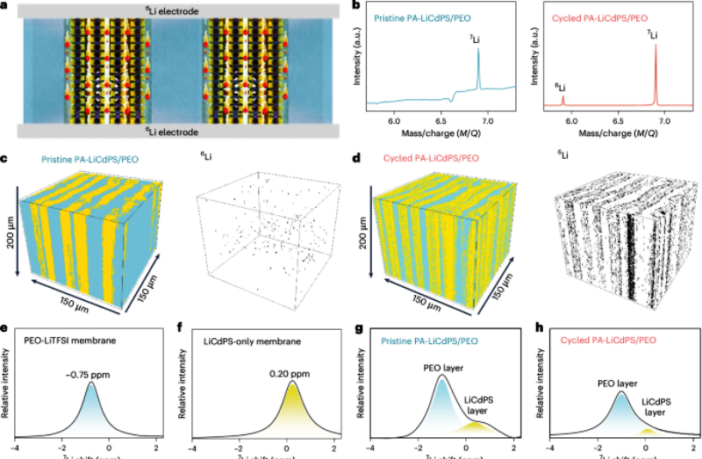
The Shenzhen Institutes of Advanced Technology came up with a novel solution, for forming a solid electrolyte as fast as liquid. In simple terms, they divided the solid electrolyte into alternating layers of the two materials.
They had found a way to resolve the twin challenges independently! They simply separated the flow of ions from the need for a solid electrolyte-electrode interlayer.
The inorganic material created the pathways for the ions to flow. While its polymer counterpart kept the solid electrolyte flexible. It was also able to maintain close contact, as the battery expanded and contracted while it discharged and recharged.
More Information
Creating Solid Electrolytes For Batteries
Solid Electrolyte Interface Trips Lithium-Metal