The media are awash with tales of electric vehicle owners queuing for hours in North America to recharge their batteries this winter. It’s an unfortunate fact that batteries recharge more slowly at lower temperatures. Perhaps we should ask electric vehicle manufactures why this comes as such a surprise. Especially since the formula for temperature batteries and the Arrhenius equation goes back to 1889.
Arrhenius’ Chemical Formula for Batteries and Temperature
Svante August Arrhenius was a founder of physical chemistry, and the first Swedish winner of the Nobel Prize. He was also the first person to determine how increasing atmospheric carbon dioxide is raising Earth’s surface temperature.
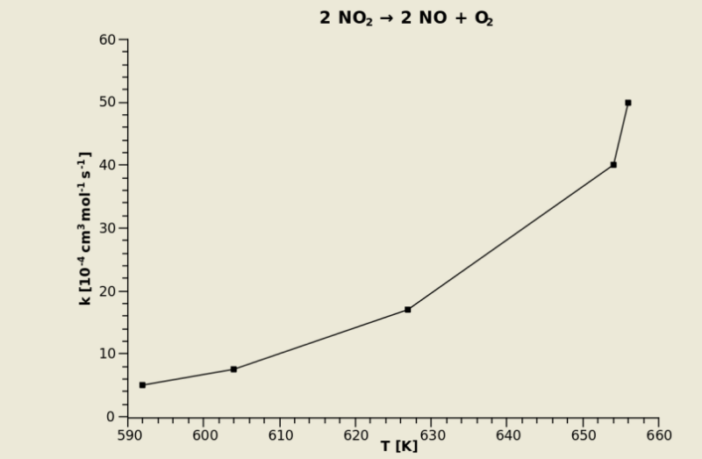
Arrhenius determined that chemical reaction rates are dependent on temperature. Without delving too deeply into the science (there is a link to a Wikipedia article below) the frequency of collisions (K) resulting in reactions, increases rapidly with rising temperature (T) per our image above, but the opposite also applies.

A Layperson’s View of the Arrhenius Equation
We are thankful to be over with chemistry, and able to return to our world of batteries. But first, let’s recap on what we discovered. We learned as kids how temperature affects the speed of chemical reactions:
- Milk turns sour more rapidly at room temperature.
- But butter stays fresher longer if we keep it in the fridge.
- Soup boils more rapidly as we turn the heat up on high.
Increasing temperature causes the molecules to move faster, and collide more vigorously, according to Libre Texts. By the same token, they will move more slowly, and collide less vigorously at lower temperatures.
Implications for Temperature, Batteries and Charging
All electro-chemical batteries do their work, and produce electricity during a chemical reaction. Therefore, their recharging rates will be likewise affected. This is why electric vehicles take longer to charge in winter. And for that matter our phones and laptops, when we are outside.
More Information
Electrolytes in Electro-Chemistry Moving Ions
Lead-Acid Battery Assembly and Chemistry