Magnesium batteries traditionally use magnesium metal, as opposed to lithium and sodium as their charge carriers, and sometimes in their anodes too. Primary, single-use versions have had some commercial success as reserve and primary-use batteries. However, until now, research has not delivered practical secondary versions. Today, we bring news of a magnesium rechargeable battery discovery, that might just knock lithium off its perch.
Practical Magnesium Rechargeable Battery From KIST
A team at Korea Institute of Science and Technology (KIST) has been chasing this possibility for a while. And why not, seeing as Wikipedia confirms that magnesium might replace lithium-ion in ‘certain applications’. KIST’s achievements from this latest discovery include:
- Increased charge and discharge efficiency, through a ‘magnesium metal chemical-activation process’.
- A compatible ‘non-corrosive general electrolyte’ that could lead to commercial secondary batteries.
KIST’s magnesium rechargeable battery could become a viable alternative to lithium ion technology, if the following facts pan out in their favor:
- Demand for lithium-ion batteries continues to expand to meet electric transport and storage requirements.
- However, the supply of critical lithium and cobalt minerals is restricted to a few specific countries.
- Concerns are growing that this unstable situation could worsen due to current international tensions.
What This Situation Could Mean for Our World of Batteries
A rechargeable magnesium battery should have high energy density. This is because magnesium, unlike lithium, has a divalent ion, capable of forming two bonds. In fact, using the metal as anode produces a volumetric density almost twice lithium’s.
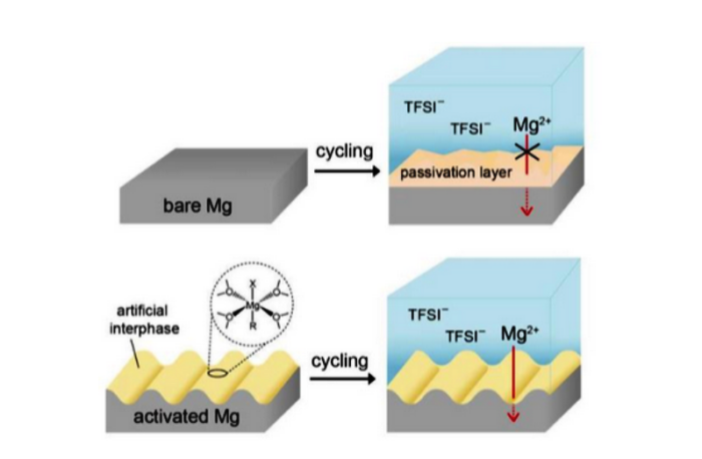
However, and this has been the catch with secondary magnesium batteries until now, they have been stubbornly difficult to charge and discharge. This problem has inevitably boiled down to their high reactivity with electrolytes.
The KIST team appears to have solved this problem with their magnesium rechargeable battery discovery. In simple terms, they applied an artificial protective layer to the magnesium surface, after dipping the anode in a novel solution. This suppressed the unwanted reactions with the electrode, enabling their work to move forward.

Magnesium Anode Dipped in Plating Solution Has Same Capacity As Larger Lithium Anode (Image KIST)
More Information
Magnesium-Ion Battery Chemistry Makes a Bid
Magnesium Ions Dance Through Solid Electrolyte