Researchers at Tongji University, and Huazhong University of Science investigated stresses in lithium-metal batteries. This was a departure from the traditional electrochemical approach, because it probed the physical resilience of the materials themselves. This line of reasoning may help explain premature solid-state lithium-metal battery failure. But first, how are these lithium-metal batteries different?
The Lithium-Metal Batteries Experiencing Stressors
Secondary, rechargeable lithium-metal batteries, use the metal for their anodes instead of traditional graphite. This novel combination delivers extremely high theoretical storage capacity, teamed with low density that outshines alternatives.
A very low negative electro-chemical potential completes a combination that could make it a winner. However, solid state, rechargeable lithium-metal batteries suffer from early battery failure, that has hindered large-scale, practical applications.
The researchers, writing in the Journal Science that we link to below, confirm that lithium-metal batteries may short-circuit due to dendrites forming on the solid electrolyte interphase layer.
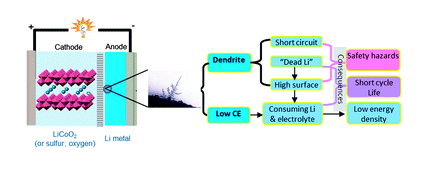
There is, however, a second cause of lithium-metal battery failure, namely low couloumbic efficiency (CE) during lithium depositing and stripping. This term refers to how effectively a battery charge transfers, during charging and discharging cycles.
The researchers applied their minds to better understanding the stresses in lithium-metal batteries affecting this phenomenon.
Metal Fatigue at the Heart of Lithium-Metal Batteries
The researchers from Tongji University in Shanghai, and Huazhong University of Science in Wuhan, China, made a lateral-thinking discovery. They established that solid electrolytes have difficulty absorbing stresses, caused by lithium contraction and expansion during charging cycles.
This phenomenon can cause dendrite formation, or even cracking as Xinhua News Agency explains. The researchers conclude that this ‘cycle fatigue’ is akin to bending a paperclip, until it weakens and finally breaks.
More Information
Solid Electrolyte Interface Trips Lithium-Metal
Resting a Battery Restores Lithium-Metal




