Lithium metal batteries outperform their competition, because their anodes can store more energy. But lithium’s ability to interact with any material is proving to be its achilles heel in this regard. That is because lithium metal dendrites form at the liquid electrolyte, to an extent that endangers the battery. Today, we report on progress made towards understanding lithium dendrites threads better.
How Electrolyte Composition Affects Lithium Dendrites Threads
Non-flammable, solid electrolytes in lithium metal batteries eliminate the likelihood of fires, but not overheating. Composite electrolytes of polymers and ceramic particles are a promising opportunity in this regard.
Scientists from the Department of Molecular Chemistry and Materials Science, at Weizmann Institute of Science in Israel, decided to look deeper into this option. They were particularly interested in the ideal blend of polymers and ceramic particles for long battery life.
Understanding the Process Leading to Electrode Short Circuiting
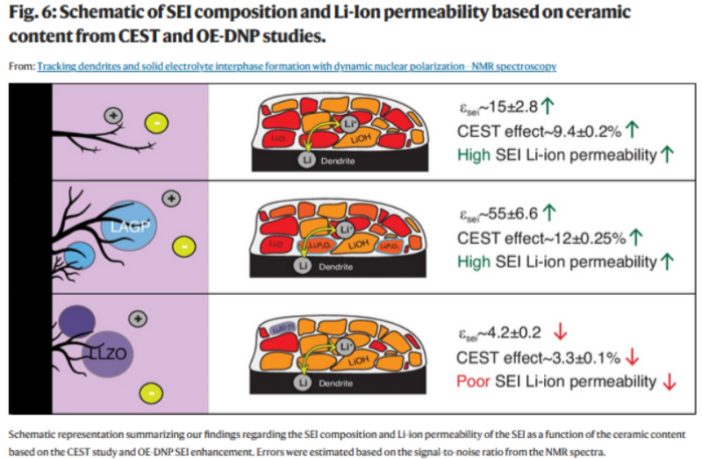
The Weizmann Institute team decided to follow dendrite development, in order to identify chemical interactions within the electrolyte. They used a technique called nuclear magnetic resonance spectroscopy to observe dendrites in real time, and relate this to chemical interactions within the electrolyte itself.
The researchers experimented with different proportions of polymer and ceramic in lithium metal battery electrolytes. They advise their findings in the report we link to below as follows:
- Batteries with 40% ceramic in their electrolyte had the longest operating lifespan.
- Structural and functional problems reduced battery performance above 40%.
- While lithium metal battery operating life began shortening below 40%.
- But the best performing batteries developed more dendrites than those outside 40%.
- However, this dendrite growth was stunted, and so they penetrated less frequently.
The team quite naturally wondered what was impeding dendrite growth in the best-performing 40% ceramic batteries. They surmised thattheir answer lay in the solid electrolyte interphase layer (SEI). We’ll pick up the threads in this in our next post.
More Information
Solid Ceramic Electrolytes For Our Future
Increasing Lithium Battery Lifespan in Lab