A redox flow battery converts chemical energy into electrical energy, through reversible oxidation and reduction of separate liquids. This is what sets their chemistry apart from conventional batteries, where this process occurs at electrodes. Scientists in China have developed a new catalytic electrode for flow batteries that improves their efficiency.
Improving Flow Battery Efficiency With New Electrodes
Round trip battery efficiency is the ratio between the amount of electricity stored from an external source, to the amount of energy the battery delivers during discharge.
No battery is 100% efficient in this regard, however even the best flow batteries only achieve 80%. None the less, redox flow batteries are popular for bulk energy storage, because some may repeat the cycle up to 10,000 times.
Flow batteries use their electrodes to connect with external devices, to charge and discharge their energy. A new catalytic electrode for flow batteries from China improves the efficiency of this interface.
More Detail About the Catalytic Flow Electrode
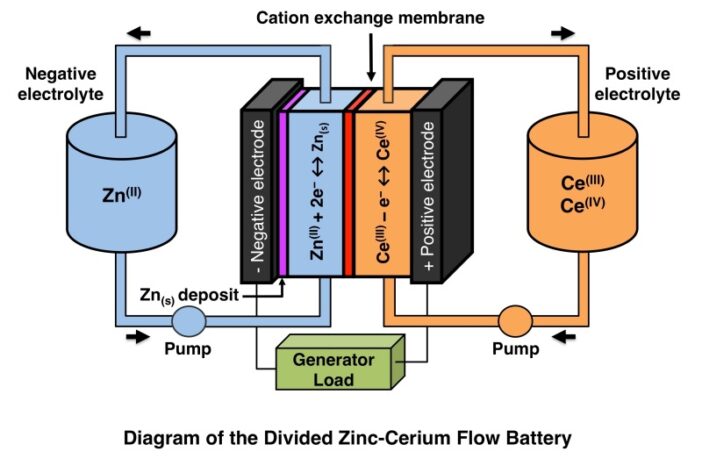
There are many different redox flow battery chemistries, including the zinc-cerium type we illustrate in our image.
In this instance though, the Chinese researchers used a polysulfide-iodide redox flow battery (SIRFB) for their experiment, perhaps because this chemistry shows potential for low cost, high energy density, and scalability:
- First, they obtained a two-dimensional sheet of inorganic molybdenum disulfide(MoS2).
- Then they enhanced the MoS2 sheet with carbon monoxide (CO) that generated abundant sulfur (S).
The new material they created (CoSA-VS/MoS2) delivered a catalytic electrode that enhanced the performance of their polysulfide-iodide redox flow battery. It achieved 87.9% round trip efficiency when tested under laboratory conditions. However, it was only able to cycle 850 times.
More Information
Redox Flow Batteries Could Come of Age
Flow Batteries for Long Energy Storage