Conventional lithium-ion batteries can perhaps go no further in terms of their performance, according to Science |Business. That’s because their graphite anodes can only store a limited number of ions. A team from Federal Institute for Materials Research and Testing in Berlin, hopes to resolve this shortfall with a new liquid alkali metal anode in a solid-state battery.
Why Do We Need a Liquid Alkali Metal Anode Now?
Researchers have been trying to find an alternative anode for solid-state batteries for a number of years, but with only limited success. The team from Federal Institute for Materials Research and Testing first considered pure lithium, or perhaps even pure sodium. That’s because either of these two options show potential to increase energy density by up to 40%.
However both these ideas have a catch, as often happens in the world of battery research. Either of the pure metal anodes must partner with a solid electrolyte for the idea to work. But unfortunately that arrangement would result in ‘contact losses and voids, rendering the battery useless.
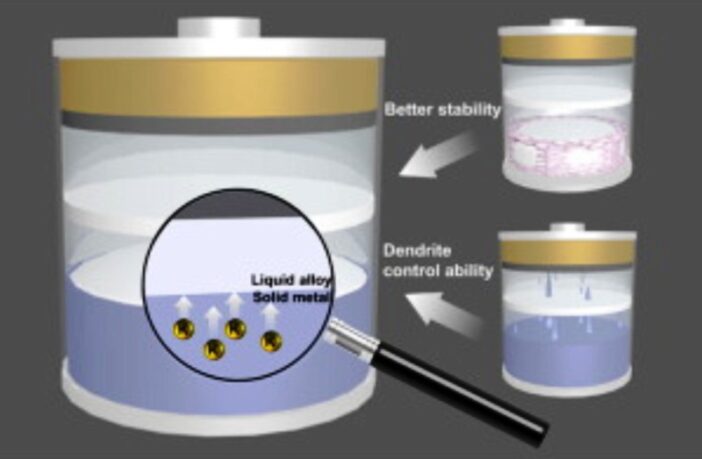
And so the researchers decided to explore the alternative of a partially liquid anode instead, in conjunction with a solid electrolyte. This option typically has solid and liquid phases, with a solid core and a thin layer of liquid alloy on top. Benefits may include improved stability, dendrite suppression, and increased capacity too.
An Alternative 100 Times More Powerful Than Graphite
“We were able to show that a liquid alkali metal anode is 100 times more powerful than a conventional graphite anode,” a team member explains. “However, this technology can currently only be used at 250 degrees celsius,” which makes it impractical”.
The answer could lie in lowering the melting point of a new liquid alkali metal anode. The researchers are currently exploring potassium additives. However again there’s a catch, because many solid electrolytes become unstable when they make contact with potassium.
However, the Federal Institute for Materials Research and Testing team has not given up. They are now experimenting with sodium super-ionic conductors (NASICON), that tolerate potassium doped with the silvery gray transitional metal hafnium.
This material is rare and expensive, but sodium solid-state batteries could drastically reduce charging times. And this should in turn significantly improve the performance of mobile and stationary energy storage systems. Just imagine an anode material 100 times more powerful than graphite!

More Information
Thin-Film Coatings on Solid State Batteries
Anode-Free Solid-State Batteries At Princeton
Preview Image: A Sample of Hafnium Metal