A lead acid battery converts the chemical energy in its active materials into electrical energy, during a chemical reaction. Although it usually comprises several identical cells to increase the output voltage. This is the first in a short series summarizing the basics of lead acid batteries. There is more to follow in subsequent articles.
Basic Working Principles of Lead Acid Batteries
All electrochemical batteries follow the same basic principles. These are (a) we can store electrochemical energy in them, and (b) we can retrieve it later. However, more advanced batteries allow us to replenish them too. Did you know lead acid batteries were the first commercially-successful design, and their users could recharge them from the beginning?
Core Components of Lead Battery Cells
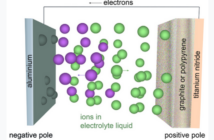
- The negative electrode is sponge lead (Pb) when in a fully-charged state.
- The lead dioxide (PbO2) positive electrode accepts electrons during discharge.
- This dissimilarity in materials allows this electric potential to materialize.
- The chemical energy becomes electrical energy when we connect the terminals.
- Meanwhile the electrolyte forms the internal circuit allowing exchange of ions.
- A porous separator prevents physical contact between the two electrodes.
Normative Cell and Battery Ratings
The potential difference between electrodes determines the voltage of a battery cell. In the case of lead acid batteries, this basic measure is nominally 2 volts per individual cell. While a full discharged one will read approximately 1.75 volts. Hence a three-cell battery should rate between at least 6, and 5.25 volts depending on state of charge.
Lead acid batteries have a well-established role in energy storage, because they are relatively cheap in return for reliable power. Never open one up out of curiosity for what is inside. They contain an acid-based electrolyte that burns human skin and eyes, and they can deliver a hefty electric shock.
This completes our first article covering the basics of lead acid batteries. We’ll be back after a short while to share thoughts about capacity and battery ratings. Then we’ll move on to explore series and parallel connections in more detail.
More Information
Barocaloric Effect in Thermal Batteries