Bipolar batteries use an electrode that is both an anode and a cathode. This potentially increases their efficiency, although a delicate balance is not easy to achieve, according to the website One Charge. We explore the basics of the mechanics and chemistry, and how bipolar battery dual-purpose electrodes work.
Dual-Purpose Electrodes in Bipolar Batteries
Bipolar batteries are elegantly simple, at least in theory. The positive and negative electrodes in a cell position back-to-back, on an underlying conductive substrate. This substance also functions as the separator, thereby electrically isolating the cell from adjacent ones in the battery.
Electric current flows directly between the bipolar battery dual-purpose electrodes, and adjacent electrodes via thin layers of conductive substrate. This eliminates the need for inter-cell jumpers. Chains of bipolar cells are in individual modules, which combine to form a bipolar battery.
To summarize at this point in the description:
- A bipolar battery comprises a number of cells in a serial arrangement.
- These cells are separated from each other by a thin, conductive substrate.
- Electrical current flows between the cells without needing mechanical jumpers.
This arrangement simplifies cell assembly, and reduces manufacturing cost, while improving energy density in a lightweight battery pack.

Bipolar Battery Electro-Chemistry in a Nutshell
Thus far we have shared the electro-mechanical structure of bipolar batteries. Now it’s time to turn our attention to the electro-chemical principles driving these:
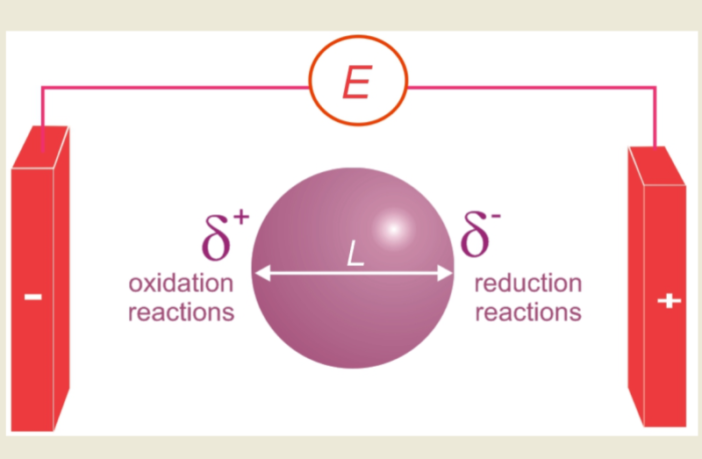
- Polarizing conducting objects in electric fields, generates potential differences between them.
- The potential difference is equal to the electric field value, multiplied by the size of the object.
- If this difference is sufficient, then redox reactions generate at the extremities of the object.
- Oxidation occurs at one extremity, simultaneously coupled to reduction at the other extremity.
If we were to place an electrically conductive material between an anode and a cathode, in the same electrolyte, and applied sufficient voltage, then that material would experience simultaneous cathodic and anodic reactions at both extremes.
In other words, the conductive material becomes a bipolar electrode in contact with an ionically conductive electrolyte, but with no direct electronic connection to the power supply. Hence it becomes and anode and a cathode at the same time, completing our introduction to bipolar battery dual-purpose electrodes.
More Information
How A Gain in Electrons is a Reduction
First Bipolar Battery Had Lead Plates