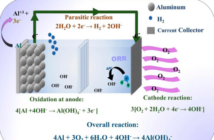
Metal-air batteries are back on the agenda, because with the right choice of metals they outperform lithium ion. We wrote recently about a new initiative using iron. However, their cells are quite bulky. So we decided it would be more practical to build an aluminum-air battery at home.
Materials for the Aluminum-Air Battery
- Some new aluminum kitchen foil
- A quantity of clean, cold tap water
- A quantity of kitchen table salt
- Some new, clean blotting paper
- A quantity of fine charcoal dust
- Red electric wire with bare ends
- Black electric wire with bare ends
- One light emitting diode (LED)
- A responsible adult to stand by
A Slightly Different Way to Achieve the Same Goal
Now Let’s Get Back to Our Experiment
First, create a saturated solution of water and salt. Then use this liquid to soak a piece of blotting paper. Gently lift this, and place it on top of a slightly larger piece of aluminum foil. Make sure it stays clear of the foil edges, as you build an aluminum-air battery.
Now sprinkle a quantity of fine charcoal dust over the blotting paper, but do take care to keep the outside edges some 10% clear. Then lay the red wire over this with one bare end contacting the charcoal dust, and the other end clear of your work.
Next, place a second, similar-size piece of saturated blotting paper on top of the first one, so you cover your work with the exception of the aluminum foil edges. Then tape the black wire in position making sure the bare edge touches the second piece of blotting paper.
As you do so, ensure the other end of the black wire is within a short distance of the visible end of the red one. Finally, complete the circuit by connecting the light emitting diode (LED) between those two terminals so it lights up. You have just built an aluminum-air battery, and demonstrated it. Congratulations!
More Information
Iron-Air Batteries on Watch List
Prove the Principles of Electromagnetism