A fuel cell is an electrochemical device something like a battery, but there are significant differences. In a nut shell, fuel cell basics combine hydrogen fuel with oxygen to produce electricity, heat and water. This process continues until the cell uses up all the fuel. Although an operator could top it up with more fuel and continue the process.
Fuel Cell Basics Ensure Zero Harmful Emissions
The hydrogen stores in a pressurized container, while the oxygen source is pure fresh air. The only by-product is the water, and there’s a virtually unlimited use for that! The hydrogen fuel cell basics that follow are the reverse of electrolysis, whereby we use electricity to split water into hydrogen and oxygen.
- The fuel cell anode, or negative electrode receives the hydrogen.
- The fuel cell cathode, or positive electrode collects the oxygen.
- A catalyst at the anode separates the hydrogen into ions and electrons.
- Oxygen ionizes and travels though the electrolyte to the anodic compartment.
- This oxygen combines with the hydrogen already in that compartment.
- Several fuel cells may connect in series to increase the total voltage.
That Sounds Expensive. Are Fuel Cells Effective?
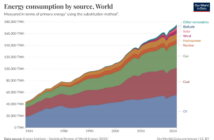
Hydrogen fuel cell technology is twice as efficient as converting carbon fossil fuels into energy. Hydrogen and oxygen are all around us. In fact, hydrogen is the third most plentiful element on Earth’s surface. Applying fuel cell basics commercially would deliver abundant green electricity, but there is a catch.
We need energy to free hydrogen from its sister substances. In fact, this would take almost as much energy as fuel cells produce. At best, we could say hydrogen fuel cells are energy neutral, and so there is no real net energy gain. The gas also requires pressurized, insulated storage. We discuss alternative fuel cell varieties in a future post.
More Information
Better Solid State Batteries with New Pathways