IMDEA Materials Institute is part of the Madrid Institute for Advanced Studies. The overall intention of the brand is to establish advanced research and higher education and training centers, in the autonomous Community of Madrid. The world of batteries watches with interest as IMDEA creates smart electrolyte for lithium-ion (li-ion) batteries. This one could be a winner!
IMDEA Smart Electrolyte Stops Li-Ion Thermal Runaway
Thermal runaway, in which a battery fire spreads across cells, could spell the end of lithium-ion chemistry eventually. But IMDEA Materials Institute has an plan to stop thermal runaway, with an ingenious heat-responsive electrolyte solution.
Previous industry efforts focused on temperature-responsive tri-layer polypropylene / polyethylene separators. However, this idea foundered because it only proved useful up to 160°C (320°F), due to thermal shrinkage.
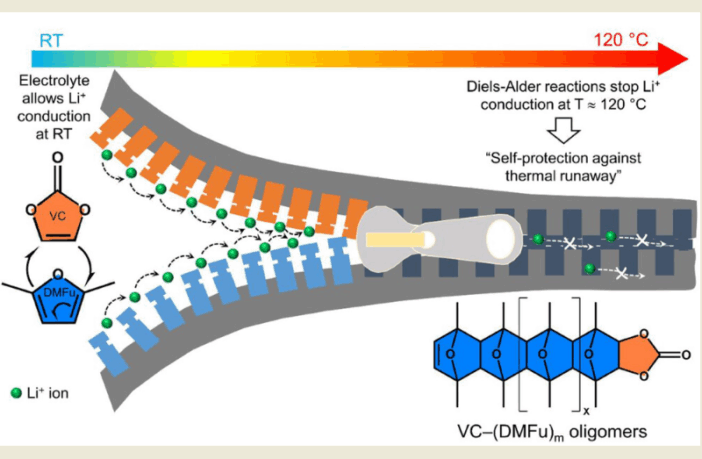
The team at IMDEA Materials Institute, under guidance of Dr. Arnab Gosh, decided to explore the potential of temperature-sensitive electrolytes instead. They hoped to design one which would block lithium ion transport, when the electrolyte warmed above 100 to 120°C (212 to 248°F).
Blocking the ongoing transport of lithium ions this way, should limit internal temperature increases, and reduce the risk of catastrophic failure due to thermal shrinkage of the tri-layer separators. We wish we could watch a video as IMDEA creates smart electrolyte live, but we do have the research report to hand (see link below).
How the IMDEA Team Plans to Stop Li-Ion Runaway
The IMDEA team developed an electrolyte composed of a lithium salt, dissolved in vinylene carbonate and 2,5-dimethylfuran:
- Vinylene carbonate is a popular lithium-ion electrolyte. It helps create a barrier between the electrolyte, and the negative anode electrode.
- The 2,5-dimethylfuran compound is a potential competitor for gasoline. It also serves as a flavoring agent, and food preservative.
This is a strange combination indeed, but the facts speak for themselves. The novel compound significantly reduces the flow of ions through the electrolyte at high temperatures, while also blocking the micro-pores in the separator.
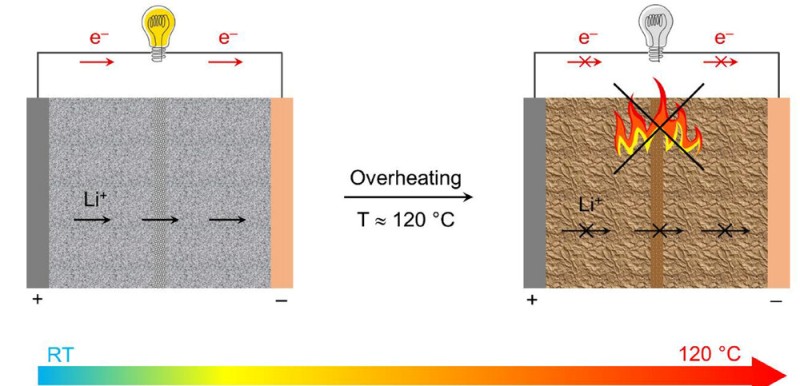
“These dual effects allow for a two-step smart regulation of thermal runaway,” a team member explains. “It enters a warning phase at 100°C (210°F), and achieves complete shutdown at 120°C (248°F). This offers a built-in safeguard against fire hazards of lithium-ion batteries.”
More Information
Upward Spiral of Battery Thermal Runaway
Thermal Runaway in Lithium-Ion Batteries
Preview Image: Illustration of Safe Battery Design