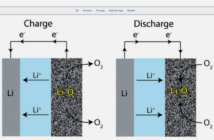
In our previous post that we link to below, we discussed what thermal runaway is, and how it may begin. We explored how the underlying cause is heat, but this may not always spread because it depends on the battery. Now we move on to describing the upward spiral of battery thermal runaway, if we are unable to stop it.
The Battery Environment And Spiraling Thermal Runaway
If the immediate environment can absorb the heat from the rogue chemical reaction, then the battery should remain safe. However, the upward spiral of battery thermal runaway may continue, if this autonomous safety factor does not kick in.
A ‘snowballing sequence’ may follow, as Gasmet Technologies puts it. ‘Heat generation increases temperature, speeds up the reaction, and generates more heat’ in a self-sustaining cycle.
This positive feedback increases the lithium-ion battery temperature extremely rapidly. The heat can soar to 600º C / 1,100º F in a matter of minutes, causing the lithium-ion battery to catch fire, and even explode.
Suppressing the lithium-ion fire, and eventually extinguishing it can take days. Conventional methods may be ineffective, because the battery chemicals generate oxygen making the fire self-sustaining. But there is more to the dilemma than that.
Gases Released During Lithium-Ion Battery Runaway
A lithium-ion battery fire does more than just release self-sustaining oxygen. It also emits a cocktail of gases, some of which are flammable while others are toxic. Gasmet Technologies mentions acids, inorganics, carbonates, volatile organic compounds, and carcinogens in the mix.
It follows that the air in the immediate vicinity may be toxic. But the actual load depends on the size of the battery. A single-cell lithium-ion fire may require no more than opening doors and windows. Whereas a burning community battery will probably necessitate greater precautions.
More Information
Thermal Runaway in Lithium-Ion Batteries