Developing the lithium-ion battery was far from being the flash of inspiration that occurred with earlier innovations. In fact it rolled out over several decades, with each contributor playing their part. Some like John Goodenough are almost modern folklore heroes. While others including M Stanley Whittingham father of intercalation seldom appear in popular literature.
M. Stanley Whittingham and Intercalation
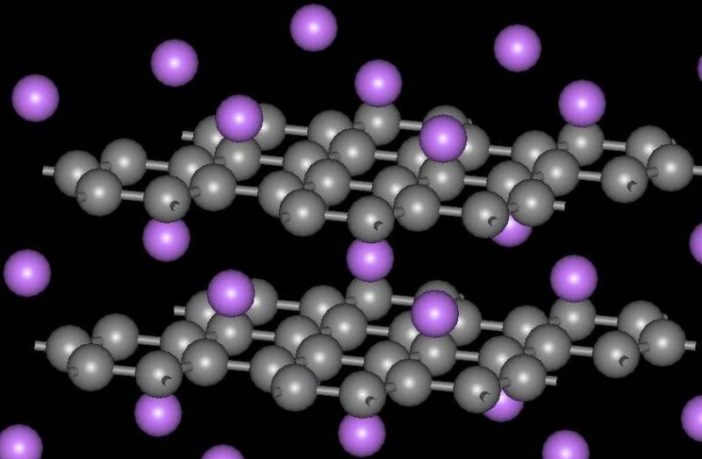
Intercalation in chemistry describes the reversible insertion, or inclusion of a molecule or ion in a layered material. This is a key process in ion exchange, while charging and discharging batteries. We take it for granted nowadays, but M Stanley Whittingham had to discover it first.
Our battery personality for today is currently a professor of chemistry at Binghamton University, State University of New York. He received the Nobel Prize for Chemistry, alongside Akira Yoshino and John B Goodenough in 2019. However, his contribution to battery history began much earlier.
That’s because he discovered intercalation electrodes in the 1970’s, and thoroughly described their role in rechargeable batteries. In fact, he still holds the original patents for conceiving intercalation chemistry in high power-density, highly reversible lithium-ion batteries.
But that was not all, by far. M Stanley Whittingham also invented the world’s first rechargeable lithium metal battery, and patented it in 1977. Although EXXON took the concept to market to energize small devices, and electric vehicles.
Was He Founding Father of Lithium-Ion Batteries?
It is not easy, perhaps unfair to single out one person for that accolade. However, he did go on to develop a practical example of his theory of intercalation in tandem with Exxon. This was a lithium-ion battery with a titanium disulfide cathode, and a lithium-aluminum anode. And it boasted high density and reversible diffusion of lithium ions.
“All these batteries are called intercalation batteries,” he once explained. “It’s like putting jam in a sandwich. You have a crystal structure, and we can put lithium ions in, take them out, and the structure’s exactly the same afterwards.
“We retain the crystal structure. That’s what makes these lithium batteries so good, allows them to cycle for so long.” M Stanley Whittingham was elected to National Academy of Engineering in 2018 “for pioneering the application of intercalation chemistry for energy storage materials.”
More Information
Lightweight Lithium Metal Takes Center Stage