Producing less carbon dioxide, and removing it from our atmosphere are two of the greatest challenges facing humanity. Metal–CO2 batteries could help us tackle these pressing issues simultaneously. Although we seldom hear of this chemistry beyond the walls of academic institutions.
A Short Introduction to Metal–CO2 Batteries
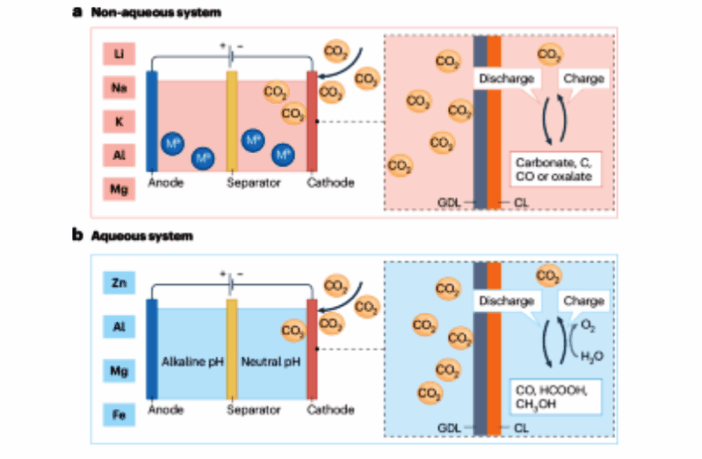
Metal–CO2 batteries are a category of metal-gas batteries, that use carbon dioxide (CO2) as active material at their cathode. This natural, colorless, and odorless gas may come directly from our environment, or from storage. The anode is a reactive metal such as lithium, manganese, aluminum or zinc etc.
This chemistry could help us rid our atmosphere of carbon dioxide greenhouse gas, which is causing global warming. This is how these batteries work:
- The reactive metal anode releases electrons as the battery discharges.
- The CO2 cathode receives these electrons and forms a stable compound.
- A liquid or solid electrolyte simultaneously allows metal ions to exchange.
Metal–carbon dioxide batteries have greater energy density than lithium-ion, because the carbon dioxide gas is light and transient. However, researchers are encountering challenges with recharging these batteries efficiently.
Promises and Realities of Carbon Dioxide Chemistry
Researchers at School of Chemical Engineering, University of Adelaide in Australia, published a paper on September 9, 2025, which we link to below. They title their work “The Promises and Reality of Metal-CO2 Batteries” which sums up the situation neatly.
Their publication compares performance, cost, and safety in aqueous and non-aqueous batteries. Here are some of their broad conclusions:
- Metal–CO2 chemistry uses carbon dioxide to offer efficient energy storage, while contributing to global net-zero goals.
- Electrode architecture, electrolyte formulation, anode engineering, separator optimization, and gas purity affect battery stability.
- The non-aqueous variety delivers high energy storage. The aqueous version converts CO2 into value-added chemicals.
- The performance of both battery types depends heavily on the purity of carbon dioxide gas. They need greater tolerance to impurities.
We wish the team at School of Chemical Engineering, University of Adelaide every success with their project going forward!
More Information
Surrey Battery Captures CO2 Gas
A Mars Battery For a CO2 Rich Atmosphere