We could record our daily rainfall, or string our readings together to determine a pattern. Quantitative mapping does something similar in chemistry. It uses numerical data to create topic maps, revealing trends and relationships between materials. Scientists at Contemporary Amperex Technology Co. Limited in Ningde, China (CATL) have made a breakthrough, after quantitatively mapping lithium-ion batteries.
Quantitative Mapping For Long, Dense Battery Life
The CATL media release that we link to below, takes lithium-ion battery research into previously unknown territory. That’s because it makes high energy density and extended battery life simultaneously possible, resolving a long-standing tension.
A Fresh Approach Opens New Doors for Li-Ion Batteries
This finding extends beyond the antiseptic walls of laboratories, where much battery research ends. In this instance though, the media release confirms that a lithium-ion prototype has achieved an energy density of over 500 watt-hours per kilogram, despite 483 discharging-recharging cycles.
This finding, ‘marks a significant step toward commercial viability for applications like electric vehicles and electric aviation’, the report continues. We explore how CATL achieved this in the following paragraph, and how quantitatively mapping lithium-ion chemistry contributed to this success.
Quantitatively Mapping Lithium-Ion Chemistry
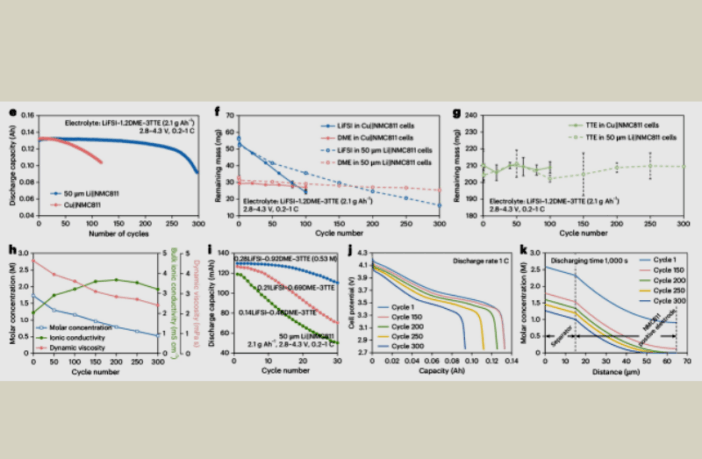
Previous efforts to overcome the tension between lithium-ion density and cycle life have been less successful, according to CATL. But they overcame this challenge through insights gained after quantitatively mapping lithium-ion active materials and electrolyte components.
The CATL team report that this method, ‘unveiled the depletion pathways driving cell failure’. However, they found that this does not occur due to ‘solvent breakdown, dead lithium accumulation, or solvation environment disruption’.
Instead, the root cause is ‘the continuous consumption of the electrolyte salt lithium bis(fluorosulfonyl)imide (LiFSI), with 71% of it consumed by end of life’. In other words, we should look beyond the effectiveness of ion shuttling, to the durability of the electrolyte itself.
CATL used these insights to dilute the electrolyte with an material of a lower molecular weight. This improved the LiFSI salt’s ionic conductivity, and reduced viscosity without increasing the total mass of the electrolyte.
More Information
CATL Promises to Share Battery Technology
CATL Super Fast Battery Challenges BYD
Preview Image: Quantitatively Mapping Pathways