An expert in the field of chemical engineering would define the fuel as hydrogen and the cells as the series of mechanical devices that convert the fuel and air (oxygen) into electricity, with heat and water as waste-products. Battery and fuel cells, despite both being electrochemical energy conversion devices, differ from one another in many aspects. One of these differences is that fuel cells generate electricity with continuous external supply of hydrogen and oxygen.
Fuel cell is a growing industry that serves the transport, stationary, and portable sectors. Fuel cells are more expensive than rechargeable batteries, but they are considered more energy efficient in the sense that waste heat produced by fuel cells is still converted into energy during operation.
Components and Operation
What makes up the fuel cells are the anode, cathode, electrolyte and catalyst. But of course, a fuel cell cannot function without the supply of fuel such as hydrogen and hydrocarbons and air (oxygen). And for this device to fully function, it will also require some external circuitry and mechanical accessories, depending on the application.
Electrochemical Reaction Process in Fuel Cells
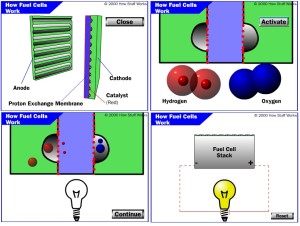
Image Source: How Stuff Works Website
A fuel cell operates via electrochemical reactions. In polymer electrolyte membrane fuel cells (PEMFC), hydrogen gas is forced in the anode while oxygen is on the cathode. The anode and cathode have etched channels to facilitate equal distribution of hydrogen and oxygen, respectively. Hydrogen splits into 2 ions and 2 electrons upon contact with the catalyst. The electrons make their way to the external circuit looping back into the cathode side in the form of direct current (DC) voltage which is in the range of 0.6V to 0.7V. Fuel cells arranged in series called stack, providing higher voltages, are what powers up the electric loads such as light and motor. The ions pass through the electrolyte (which also blocks the electrons) and then combine with the 2 oxygen atoms and 2 electrons at the cathode side, forming a water molecule.
Types and Application
PEMFC, having high power density and low operating temperature, is used primarily in the transport industry. PEMFC powers the hydrogen-powered electric vehicles which in turn rely on hydrogen refuelling stations.
Solid oxide fuel cells (SOFC) which operate at very high temperature (up to 1000⁰C) are used as an auxiliary power source and electric utility. Recent developments in SOFC make fuel cell chargers for portable consumer electronics available in the market.
A process known as co-generation of heat and power (CHP) converts the waste heat into something useful for the homes, offices and factories while at the same time benefiting from the electric current produced by fuel cells. Types of fuel cells suitable for CHP processing are molten-carbonate fuel cell (MCFC) and phosphoric-acid fuel cell (PAFC).
Many highly-funded research and development projects are on-going and they are all focusing into the same direction – make fuel cells practical and cost-effective source of energy for all of us.
Related articles:
Rechargeable vs. Single-use Batteries