Breaking up of any liquid into its chemical components by passing electricity through it is called electrolysis. Water is also a liquid and hence can be broken into its components using electrolysis. Let’s first see the chemical composition of water:
2H2O = 2H2 + O2
What this essentially tells us is that 2 molecules of hydrogen and 1 molecule of oxygen form 2 molecules of water. The hydrogen and oxygen molecules in a water molecule are held together by a force called bond. This bond is broken when electric current is applied and the molecules become free.
So we will get hydrogen and oxygen on electrolysis of water. Now we will see how the splitting of water can be done in an experimental set up.
Materials Required
You will need following materials for conducting electrolysis of water:
- Specially designed water container (as shown in Figure 1)
- Water
- 1 tsp table salt
- Two gas containers
- 12 V Battery
- Electrodes
- Two Wires

Figure 1
Experiment Procedure
Follow these steps to collect hydrogen and oxygen gases released due to electrolysis of water.
- Attach the two electrodes as shown in Figure 1.
- Fill the container with water.
- Add salt to the water so that it conducts electricity.
- Attach gas containers to the ends of the two water columns.
- Connect battery to the electrodes using the wires and your set up is complete
Experiment Results
Observe gas bubbles forming over the electrodes, also called anode (+) and cathode (-). As they rise to their respective containers, the containers will expand in size. You will see that one container will be considerably bigger than the other one. Remember that water has double the hydrogen than oxygen? The bigger container has hydrogen while the smaller one has oxygen.
We have added salt to make the water conduct electricity. Its chemical composition is NaCl. So chlorine gas is also released during electrolysis, but as it is heavier than both oxygen and hydrogen, it will not rise to the gas containers.
Simpler Experimental Set up
If you feel the set up described above is cumbersome for you, you can make an alternate set up to demonstrate splitting of water using electrolysis. Here is what you need to do:
- Fill a glass with 2/3rd water.
- Add a pinch of salt.
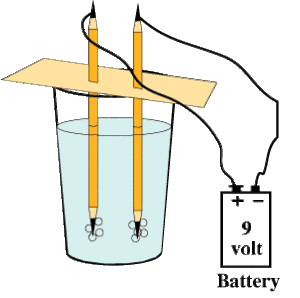
- Sharpen two pencils on both the ends.
- Pass the pencils through a cardboard and cover the glass with it, as shown in Figure 2.
- Attach the lower ends of the pencils to a 9V battery using wires. Your demonstration is ready.
Figure 2
The pencil leads act as electrodes in this experiment. Observe gas bubbles forming around the two pencil ends. Here you will also get chlorine gas with hydrogen and oxygen in water itself.
The hydrogen and oxygen gas are produced at industrial levels through electrolysis. After filtration oxygen can be used in hospitals while hydrogen can be used as fuel.
Related articles:
Anode