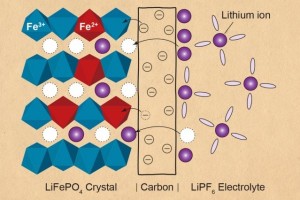
A recent finding in lithium-ion battery shows that it is actually the electrons (and not the ions) that are transferring from the solid carbon coating going through another solid which is the electrode. This surprising discovery that stems from experiments conducted by Peng Bai and Martin Bazant of MIT also reveals that the previous theory that the performance of electrodes was limited by how fast lithium ions are diffusing from the liquid electrolyte to the solid electrode is wrong.
Movements of ions and electrons in a Li-on Battery during Discharge
Image Source: Science Daily Website
What is its implication in the battery industry?
Intercalation and Butler-Volmer Equation
Let’s track back this process in chemistry called “intercalation” which is the reversible inclusion of ion into compounds with layered structures. Note that the participants in this electrochemical reaction in a lithium-ion battery are the negative and positive electrodes with the electrolyte providing a conductive medium for lithium-ions to move between the electrodes. During intercalation, ions move into the electrode. During the reverse process, ions move back out of the electrode.
Now, according to the Butler-Volmer equation, the electrode reaction is controlled by the electrical charge transfer at the electrode (and not by the mass transfer from electrolyte to the electrode surface, and vice versa). In other words, it concludes that the intercalation is limited primarily by how fast (or charge transfer rate) lithium ions would diffuse into the solid electrode from the liquid electrolyte, and vice versa.
These assumptions in science are what have been found out as misunderstandings in Li-on battery by this recent analysis in MIT.
Marcus-Hush-Chidsey Charge Transfer Theory
This is the equation that disproves the Butler-Volmer equation which states that it is the movement of lithium-ion from the liquid electrolyte to the solid electrode (and vice versa) the electrical charge transfer is limited. Marcus-Hush-Chidsey equation states that the main limiting factor in the electrical charge transfer is the movement of electrons between the porous solid electrode and the solid carbon coating (and vice versa). To highlight the main difference, Butler-Volmer suggests charge transfer is on “liquid to solid (and vice versa) movement of ions”, while Marcus-Hush-Chidsey concludes it is on “solid to solid (and vice versa) movement of electrons”.
What does this mean for us?
Lithium-ion battery is one of the most widely used rechargeable battery products. In 2012, the estimated sales of Li-on battery across the US was close to 4B dollars. This puts Li-on second only to lead-acid battery which has an estimated sales of 6B dollars.
The recent finding can be a major breakthrough in further improving the charge and discharge characteristics of Li-on batteries. In was cited by Peng Bai that the focus right now should be on how to engineer the surface at the solid to solid interface. In addition to that, Martin Bazant mentioned that apart from improving the electrode design, research and application can further extend to better understanding of other electrochemical processes such as electrodeposition, corrosion, and fuel cells.
Related articles: