We had not known, but now do about Recovery trials ongoing to reduce COVID-19 deaths. This is an acronym for Randomized Evaluation of COVID-19 Therapy, and the program is a University of Oxford outreach. The team there is testing suggested treatments, in hope of helping people recover from severe infections. Breaking news is they just identified a monoclonal therapy reducing COVID deaths. However, there are narrow limits to it’s application.
The First Convincing Evidence of a Working Therapy
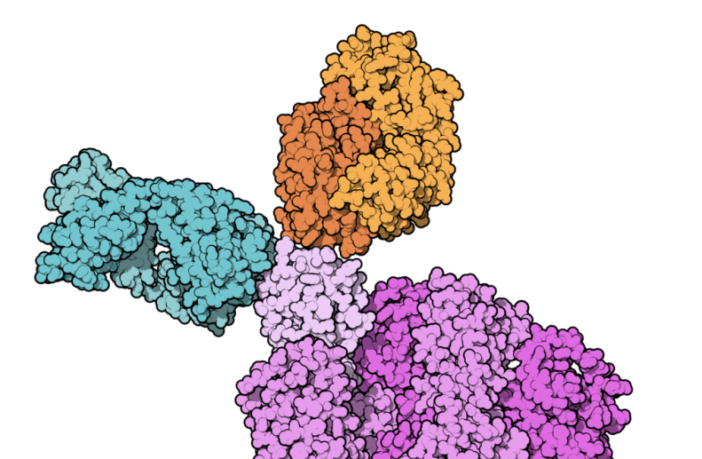
The American Association of Scientists (AAAS) advises the trials produced convincing evidence of a therapy that works. The medication is a mixture of two monoclonal antibodies manufacturer Regeneron calls casirivimab and imdevimab. Wikipedia advises the company developed the combination to reduce resistance to their monoclonal therapy now reducing COVID deaths.
Kai Kupferschmidt in Science Mag for AAAS says this combination did not produce results on all patients tested. However, it was successful in 20% of cases, where patients did not produce their own antibodies themselves. ‘Here you have really the first direct SARS-CoV-2 drug,’ says Eric Topol, director of Scripps Research Translational Institute.
But This Monoclonal Therapy Reducing COVID Deaths Has Limitations
However, Michelle Roberts is more cautious in BBC News Health. She says the potent intravenous infusion of antibodies is expensive at a minimum $1,400 per shot. And moreover it could only help a third of patients, and save lives of 6% of them at best.
Furthermore the therapy must also be administered in conjunction with the anti-inflammatory steroid drug dexamethasone. Therefore, this is hardly the time to cancel the vaccination, throw away the face mask, and leap for joy.
Nonetheless, drug manufacturer Regeneron has made progress in finding ways to help patients unable to produce own antibodies. A further problem would be rolling out the drug in developing countries in terms of cost.
Antibodies that we have benefited from in high-income countries for 20 to 30 years still aren’t available in many countries. Recovery’s Peter Horby adds ‘The inequity is a scandal. There really must be an initiative to make these drugs accessible’ to all.
Related
Why WHO Regards Delta a Variant of Concern
How Resistant Delta is to COVID Vaccines
Preview Image: Binding to the Spike Protein