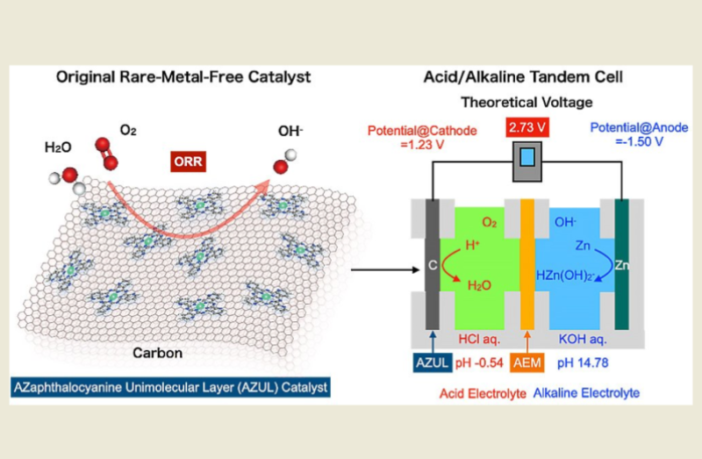
Zinc-air batteries looked good when they arrived on the scene, potentially offering high density, low-cost, and environmentally-friendly design. However, the interest fell away when it became evident the best they offered was 1.4 volts. As a result, the batteries seemed condemned to serve low power devices like hearing aids. And then along came zinc-air batteries with dual electrolyte, thanks to work at Tohoku University’s Advanced Institute for Materials Research.
Something Had to Be Done to Improve the Situation
The team who came up with the new innovation, realized that the concept required fresh thinking. That’s because zinc-air batteries needed improved drive voltage, and output power density, before they could enter the lucrative electric vehicle and drone markets. Their work has led to a zinc-air battery with an open circuit voltage of more than two volts.
Now we do know that the potential difference between anode and cathode determines the voltage a battery puts out. In the case of zinc-air batteries, dissolving zinc in the electrolyte at the anode produces the potential. While with the cathode, this is achieved by an oxygen-reduction reaction.
“We harnessed a cell with a rare-metal-free cathode and acidic/alkaline electrolytes arranged in tandem to overcome the major bottleneck for metal-air batteries,” explains Professor Hiroshi Yabu, corresponding author of the study.
Zinc-Air Batteries with Dual Electrolyte Made the Difference
From previous research the team knew the pH level of electrolyte has a marked impact on the reaction potential at each electrode. And this kept the voltage of zinc-air batteries down to 1.4 volts effectively. They investigated and discovered that:
- The dissolution potential at the anode is lowest in an alkaline environment.
- While the redox potential at the cathode is greatest under acidic conditions.
Hiroshi Yabu is team leader and professor at Advanced Institute for Materials Research, Tohoku University. He explains the higher-voltage, breakthrough zinc-air batteries with dual electrolyte as follows:
“This led us to realize that by making the electrolyte on the zinc anode side alkaline. And creating acidic conditions on the cathode side, we could generate a higher voltage.” And this in turn led to developing the dual electrolytes.
More Information
Lithium-Ion BEV Battery Forecast
Popular Types of Batteries in Use Today
Preview Image: Original and New Version
Tohoku University’s Advanced Institute for Materials Research