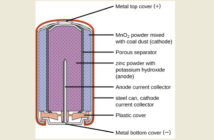
The Latin word ‘aqua’ embraces‘water’ in its many forms. Aqueous batteries use water-based solutions for their electrolytes. This means that an active material is added to the water. The energy densities and life cycles of these batteries are inadequate for grid storage and electric vehicles. However, they are safe, reliable, and inexpensive compared to lithium-ion options.
First Aqueous Batteries Used Sulfuric Acid Electrolyte Blend
The first aqueous batteries used sulfuric acid diluted with water. This reduced the cost of the electrolyte, while retaining its active properties. This invention came about thanks to the efforts of Gaston Planté, who invented the lead acid-battery in 1859.
The idea worked, and in time aluminum, carbon, lithium, manganese, potassium, and sodium versions followed. Although in the past decade lithium-ion has aggressively challenged their markets, albeit somewhat unsuccessfully. Why is this so?
Water-Based vs Lithium-Ion Batteries Who Wins?
THE ADVANTAGES OF WATER-BASED ELECTROLYTES
- Water cannot burn, in fact we can use it to put out many fires. As a result, water-based electrolytes are safe in the sense they cannot catch fire or explode.
- Lithium metal is an expensive material in relatively short supply. Many aqueous batteries use cheaper materials, for example the lead in lead-acid batteries.
- Batteries with water-based electrolytes often have faster charging and discharging rates, and are less affected by different temperatures.
THE DISADVANTAGES OF WATER-BASED ELECTROLYTES
- Batteries with water-based electrolytes have a narrow electrochemical window. And as a result only lead-acid batteries can achieve as much as two volts of energy.
- Water is an aggressive solvent that can damage battery components and cause corrosion. This restricts material choices, and the life expectancy of the cells.
Global News Wire expects the U.S. lead-acid battery market to surge from $54 billion to $94 billion by 2030. A steady development rate of 8.2% suggests they may be commoner than you think, going forward.
More Information
Zinc-Air Batteries With Dual Electrolyte