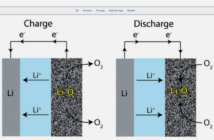
A metal-air electrochemical cell uses an anode made from pure metal, and an external cathode of ambient air. The electrolyte that completes the picture is typically either an aqueous water-based one, or a special solvent. A reduction reaction occurs at the external cathode during discharging, while the metal anode is simultaneously oxidized.
Metal-Air Electrochemical Cells Could Surprise
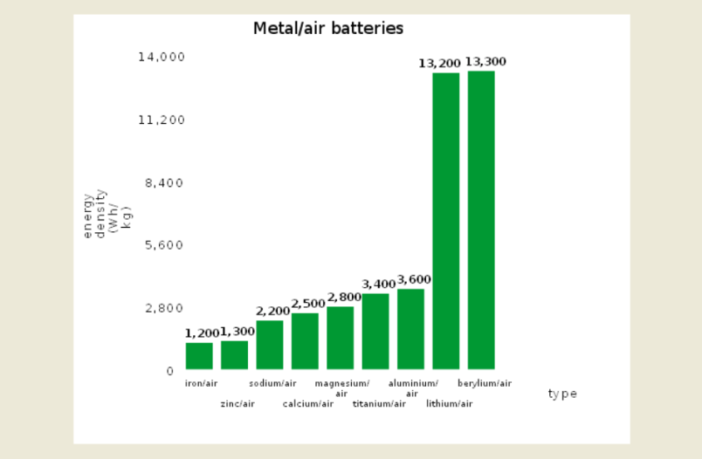
The theoretical specific capacity and energy density of metal-air electrochemical cells is higher than that of lithium-ion batteries. There are a few successful applications, such as zinc-air batteries for hearing aids. However, complications associated with metal anodes, electrolytes and catalysts have generally restricted development of metal-air batteries.
High Level Information about Various Metal-Air Electrochemical Cells
LITHIUM-AIR BATTERIES
The remarkably high density of lithium metal ensures the success of single-use (non-rechargeable) lithium-air batteries. However, incomplete discharge at the cathode precludes the possibility of secondary batteries.
SODIUM-AIR BATTERIES
There was originally hope that sodium-air batteries would overcome those shortfalls with lithium-air, even though they have less density than the latter. Some recharging has been achieved, although only up to a maximum 150 cycles.
POTASSIUM–AIR BATTERIES
Attempts were made with potassium-air batteries to achieve this same goal. However, to this day only two to three charge-discharge cycles have been achieved. None the less, the chemistry does offer an exceptionally low potential difference.
ZINC-AIR BATTERIES
Zinc-air batteries are only able to produce a maximum of 1.4 volts. Hence their usage is limited to low-voltage applications such as hearing aids, where they have an established reputation.
MAGNESIUM-AIR BATTERIES
Magnesium-air batteries have been investigated using a variety of metal chemistries. A number of drawbacks have been identified, including difficulties with aqueous electrolytes. Moreover, no successful rechargeable versions have emerged.
ALUMINUM-AIR BATTERIES
Aluminum-air batteries have the highest density of any other battery. They have potential for electric flight, because they maintain stable voltage and output until they run out of power. The lack of a metal anode also contributes to a lightweight design.
IRON-AIR BATTERIES
Iron -air rechargeable batteries are an attractive technology, particularly with grid-scale storage in mind. The main material, iron oxide which is rust, is abundantly available. The design becomes rechargeable when used in conjunction with fuel cells.
Breaking News
Will Silicon Be the Ideal Lithium-Ion Anode