Early batteries were single use, and therefore non-rechargeable. This meant their users either had to replace their materials, or discard them when their cells ran out of energy. But Gaston Plante invented a lead battery he could recharge when the cells were empty. We are unable to discover how he figured that out, but it makes for a rollicking good story.
Plante’s Lead Battery and ‘Rheostatic Machine’
Plante actually invented two things. These were his famous battery, and a mechanical ‘rheostatic machine’ producing sufficient energy to charge a bank of capacitors in parallel. We can surmise he used his rheostatic machine to recharge his own battery too, by passing a reverse current through it. But we will probably never know whether he did this on a whim, or on the basis of sound logic.
The Nuts and Bolts of the First Rechargeable Battery
A typical lead battery comprises a lead anode, and a lead dioxide cathode both immersed in sulfuric acid solution.
- The lead anode, and the lead dioxide cathode react with the sulfuric acid solution surrounding them.
- However, the lead anode releases electrons, while the lead dioxide cathode consumes these.
- This generates an electric current powering a device connected between the battery terminals.
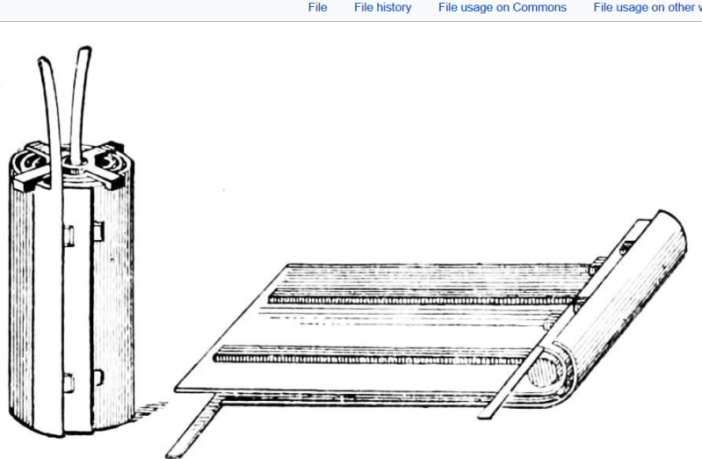
Plante’s original 1859 lead battery comprised two lead sheets, with rubber strips separating them. He rolled this into a spiral, and was able to power incandescent lights in railway carriages while stationery at a station.
But Camille Alfonse Faure derived an improvement in 1881. This comprised a lead grid lattice into which he pressed lead oxide paste, forming a plate. This made it possible to stack multiple plates, enabling greater performance and launching the technology commercially.

More Information
Experiments in Science: The Grove Cell