A lithium-ion battery gets its electricity from a chemical reaction within that silvery metal. Left to its own devices, that material could become unstable. Four parts of a lithium-ion battery control this restless energy, keeping it inside its case and managing the flow of power. Every one of these components is equally essential in the life of this remarkable device.
The Critical Role of The Cathode Electrode
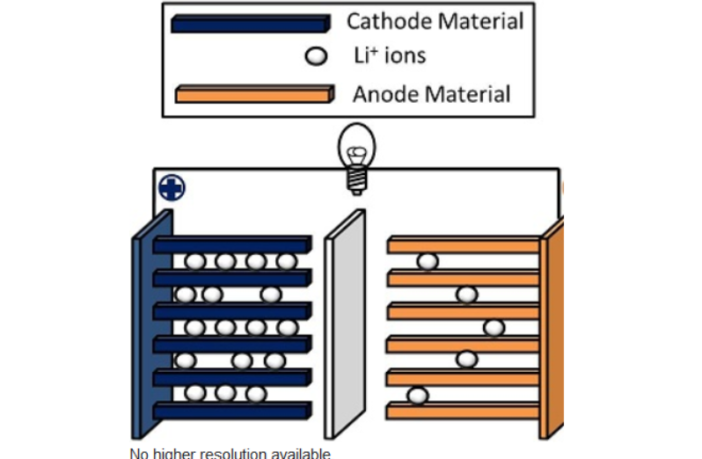
Electricity flows from a battery through its cathode electrode terminal to the device it powers. This is why the cathode in one of the critical four parts of a lithium battery, and we highlight it here.
A lithium-ion battery cathode contains lithium-oxide that coats a thin aluminum foil. This ’dilution’ keeps the lithium-ion reactions within safe limits, and so it delivers the voltage we need for our device.
The anode electrode is the second of the four components we describe here. It also has an active material coating, although this has a lower potential difference to ensure that its ions are attracted to the cathode.
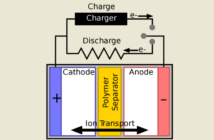
If the anode and the cathode touch directly, then all the ions will flow across in an instant. This will cause a short-circuit generating high heat. Those ions can’t flow through fresh air intervening. That’s the job of the electrolyte and separator.
The Other Two Parts of a Lithium-Ion Battery
The third part of a lithium battery is the electrolyte. This highly-active fluid allows the ions to pass through it from the anode to the cathode, and in the opposite direction when charging.
All is good with our battery with these three things in place, provided the two electrodes remain apart. The fourth component is a physical separator that keeps everything away from the electrolyte, except the active ions it allow through.
Preview Image:
Anode and Cathode Making a Perfect Pair