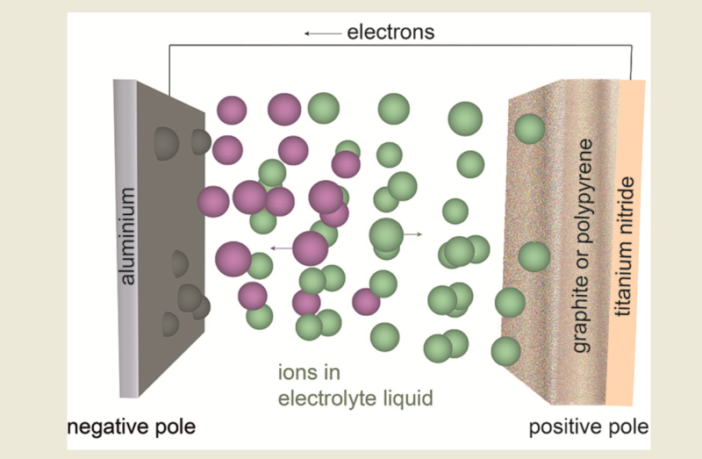
Terminals are the most obvious components of sealed electrochemical batteries. Most battery users connect them to their devices, without further thought. However, the terminals also connect to electrodes inside batteries, which control the flow of energy. Well energies really, because there are actually two different battery electrode roles.
Two Electrode Roles Depending on Battery Status
Battery electrode roles depend on the status of the battery at the time. We need to pause for a few quick definitions before continuing:
- An oxidation reaction is an electrochemical process whereby a chemical substance loses electrons to another chemical substance.
- A reduction reaction is an electrochemical process whereby a chemical substance gains electrons from another chemical substance.
- When a battery is discharging its energy to a circuit, an oxidation reaction occurs at the negative anode as its gives up electrons.
- However, at the same time a reduction reaction occurs at the positive cathode during recharging, as it receives the electrons.
More or less the opposite happens in a battery while it is recharging, because the anode and cathode flip polarities. When we apply an external current to charge a battery, the cathode (now negative) pushes the electrons back to the now positive anode to restore the state of charge.
How the Two Electrodes Rely on Different Potentials
Those two battery electrode roles that we mentioned, rely on different potentials between them to trigger the electron flow. We call the electrode with a higher potential at that point in the discharge-recharge cycle, the positive one, because it receives the electrons.
The above statement is true whether the battery is discharging or recharging. This is how the anode and cathode electrodes flip polarities during normal use of healthy batteries, and allow us to store and retrieve electrochemical energy.
More Information
Electrical Conductors We Call Electrodes