German scientist and inventor Carl Gassner invented a dry version of the wet Lechlanchė cell in 1886. This was an early, glass jar version with a carbon cathode, a zinc anode, and an ammonium chloride solution as electrolyte. However, Gassner’s zinc-carbon battery was different. He used a zinc sheet-metal case as anode, and added a plaster-of-paris gelling agent to the electrolyte.
How a Modern Zinc-Carbon Battery Works
The original Lechlanchė cell had a liquid electrolyte in a glass container making it a static battery. Gassner’s zinc casing made the chemistry portable, while the semi-solid electrolyte meant it worked upside down or on its side.
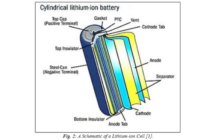
The modern version has a paper separator soaked in ammonium chloride, with a thickener added to complete an electrolyte paste. The zinc case is the negative anode, with a powdered carbon rod coated with manganese dioxide acting as the positive cathode.
Carbon is the only practical cathode in a zinc-carbon battery, because any metal electrode would corrode in the salt-based electrolyte. A separator of starch or flour completes the design of this elegantly simple battery.
The carbon rod is slightly porous, allowing more charged atoms to form hydrogen gas during discharging, which the battery contains using a thermoplastic washer seal. A higher ratio of carbon powder decreases internal resistance, while a higher proportion of manganese dioxide increases capacity.
Advantages of Using Zinc Batteries
Alkaline batteries have largely replaced zinc-based ones, because they are safer and more efficient. But zinc-carbon battery costs are lower, making them an attractive proposition for low-energy devices like clocks and television remotes.
However, these batteries have a relatively short life, due to the ammonium-chloride attacking the zinc case and corroding it. The metal case becomes thinner and thinner as it oxidizes. It eventually begins to leak sticky zinc-chloride paste, which may burn human skin and other tissues.
More Information
Why Batteries Suddenly Became So Important