An electric battery is able to store and release energy to power devices according to user requirements. It may be a stand-alone electrochemical cell, or it may incorporate one or more of these. It can switch its terminals to different roles, depending on whether it is charging or discharging. Read on and we’ll share all about electric batteries you need to know.
More About the Terminals In Electric Batteries
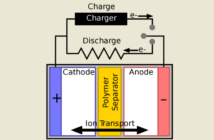
The terminals on a battery connect to electrodes inside, that store and release the energy. The following is true when a battery is discharging energy and supplying power:
- The positive terminal is the cathode, while the negative terminal is the anode.
- The negative terminal releases the electrons that flow through a circuit to the positive terminal.
- Those electrons come from a redox reaction which sends the free energy to the circuit.
That’s about all about electric batteries you need to know at this stage. You’ll find loads more information under the Basic Tab at the top of this page.
Comparing Primary and Secondary Batteries
We can only use primary batteries once, because the process permanently alters the electrodes attached to the terminals. But we could recharge secondary batteries a number of times, by sending through an electric current. This chemistry is more complex. which is why they cost more.
We use cheaper primary batteries in simple, non-essential applications like flashlights and toys. However, we use secondary batteries for important applications like auto starters, UPS systems, electric cars, laptops, smartphones and so on.
Is That All I Need To Know About Electric Batteries?
At this stage yes, because all batteries follow the same principles. But they do come in different sizes and shapes, and this can be confusing. Next time you check your smartwatch battery, remember it works the same way as the batteries that power ships, and space probes far, far away.
More Information
How A Gain in Electrons is a Reduction