We thought it would be helpful to describe the working principles of all electro-chemical cells first. Because lithium-ion batteries are a special class within those same broad principles. Every battery class is unique, because it uses a different combination of materials. So with that introduction behind us, how do lithium-ion batteries work?
The Working Principles of Lithium-Ion Batteries
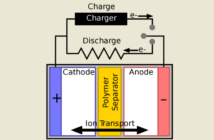
All batteries – or electro-chemical cells to be precise – transform energy from one type to another, as opposed to generating it. There are four key components in every battery, as confirmed by Argonne National Laboratory:
- A cathode comprising the positive side of the battery.
- An anode comprising the negative side of the battery.
- A separator that prevents the cathode and anode touching.
- Electrolyte allowing charges to flow between cathode and anode.
Returning to our question how do lithium-ion batteries work so well, we find the answer in their unique combination of materials:
- Lithium-ion positive cathodes comprise cobalt, manganese, and nickel oxides.
- Lithium-ion negative anodes are composed of the same graphite as many pencils.
The positive cathodes and negative anodes store lithium particles, depending on where the lithium-ion battery is in its cycle.
How Rechargeable Lithium-Ion Batteries Power Our Devices
When we turn on a device containing a lithium-ion battery that is holding charge, the ions flow through the electrolyte from the anode to the cathode. Chemical reactions follow, generating electrons. This is how these reactions convert the chemical energy in a battery into electrical current.
However, if we recharge a lithium-ion battery instead, then the chemical reactions occur in the reverse order. The lithium-ions flow from the cathode to the anode, as an external electrical current converts to chemical energy.
This cycling process repeats many hundreds of times, during the life of a rechargeable lithium-ion battery. However, each time some of the ions are lost in the process. Eventually, so many of the ions have been lost that it is time to replace the battery, and recycle the old one’s materials.
More Information
Safe Efficient Lithium-Ion Quasi-Solid Battery
How Some Lithium-Ion Batteries Fail Early