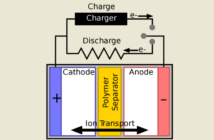
A lead-acid battery comprises a number of pairs of lead-based plates, with separators between these plates. This assembly is inside a strong plastic case, containing an aqueous solution of sulfuric acid and water. We can charge a lead battery by sending direct electric current through its terminals via a charger. Thereafter, we can discharge the battery by drawing direct electrical current through the same terminals. Does this answer your question: how does a lead acid battery work?
Want to Know More About Lead Acid Chemistry?
We did not mention the chemical processes that unfold, as we charge and discharge lead-acid batteries. We first wanted to provide a framework for the information. Here are more basics you need to know:
- The pairs of lead plates function as electrodes. The negative one is pure porous ‘spongy’ lead. The positive plate has a coating of lead dioxide.
- A liquid solution of sulfuric acid and water – or one mixed with a gelling agent – forms the electrolyte through which the plates interact.
- A separator inserts between the lead plates in each pair. This prevents them touching, so they can only interact through the electrolyte.
WHEN WE DISCHARGE A LEAD BATTERY
Discharging a lead acid battery triggers the following chemical reactions. These continue until sulfation restricts them to the point the battery is ‘flat’:
- The negative plates release electrons to an external circuit. At the same time they react with sulfate ions to produce lead sulfate.
- The positive lead plates receive electrons from the external circuit. They react with the electrolyte to produce lead sulfate and water.
- This chemistry reduces the sulfuric acid concentration in the electrolyte. Sulfation gradually insulates the plates from the electrolyte.
WHEN WE CHARGE A LEAD BATTERY
When we charge a lead acid battery those chemical reactions reverse, until most of the sulfation is gone. It is now time to stop charging the battery:
- The lead sulfate at the negative plate reverts to spongy lead. The lead sulfate at the positive plate becomes lead dioxide again.
- The sulfate ions given up during discharging return to the electrolyte. The sulfate becomes sulfuric acid, restoring the original solution.
THAT JUST LEAVES ONE QUESTION TO ANSWER …
That just leaves one question to answer: how does a lead acid battery wear out, so we need a new one? Some of the lead sulfate remains on the plates, reducing the electrolyte strength. Some of the ions never find their way home again. The voltage drops in lead-acid batteries to the point they can no longer do their work properly.
More Information
From the First Battery to Lead-Acid
Lead-Acid Battery Renewal Is Ongoing