Internal resistance in batteries causes load losses. We could compare this to an operating expense in a business, although the two are not directly related. In an ideal situation a battery would have zero resistance. In reality, all commercial batteries lose some of their ability to deliver high currents, without significant voltage drops.
Internal Resistance Defines a Battery’s Performance
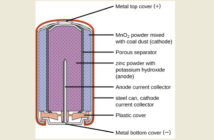
Internal resistance, without all the high-tech details, is resistance to electrical energy flow through a battery. This impedance occurs throughout the battery, including the electrodes, electrolyte and everywhere else where the energy flows.
Each of those components inside a battery causes friction, as the electrical energy passes through them. The net effect is some of that energy converts to heat. This robs the battery of some its power. More power is lost as the energy leaves the battery.
What Science Tells Us About This Loss of Power
A battery moves very tiny particles we call ions, through electrolyte between the electrodes. These ions are electron-microscopic physical pieces of the active material. Friction occurs as the tiny particles travel on their journey. There are two aspects to this internal resistance in batteries:
- Resistance as the electrical energy powers its way through the solid electrodes, tabs, and terminals.
- Resistance as the ions flow through the electrolyte. We feel something similar when we swim through water.
Both of these processes cause friction leading to heat, as the solid or liquid materials ‘rub’ up against each other.
Internal Resistance in Batteries in Practice
- The chemical structure of the battery influences the degree of internal resistance.
- When a battery is cold the electrolyte thickens, increasing resistance to ion flow.
- Batteries lose some of their energy as they age, and thin layers form on their electrodes.
- Internal battery materials experience greater strain when they are fully charged or discharged.
Internal resistance in batteries increases with ongoing use, impacting further on charging, discharging, power output, and efficiency. There comes a time in the life of every battery, when it is time to replace it.
More Information
Battery Internal Resistance and Performance