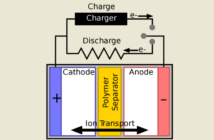
Lithium-ion batteries have several downsides resulting in restraining electric vehicle’s performance. Therefore, scientists pursue finding solutions that could increase its competence, hoping to surpass gasoline-powered vehicles.
One advancement researchers and scientists execute is through nanotechnology. Now, they aim to incorporate a powder called metal organic framework (MOF) which has the capacity to capture the polysulfide molecules, a class of chemical compounds containing chains of sulfur atoms which are formed in the cathode. Polysulfide molecules shorten the lifespan of a battery because it directs the “sulfur” into another path, losing enough materials for the energy reaction needed within the cathode. Lost sulfur are dissolved in the electrolyte, permanently eliminating an important part of the battery. With the existing issue, the MOF could be the answer to the polysulfide movement between the anode and the cathode.
“Lithium-sulfur batteries have the potential to power tomorrow’s electric vehicles, but they need to last longer after each charge and be able to be repeatedly recharged,” said materials chemist Jie Xiao of the Department of Energy’s Pacific Northwest National Laboratory (PNNL). “Our metal organic framework may offer a new way to make that happen.”
Formation of Polysulfide Molecules
A lithium-ion battery has two electrodes: the cathode which is the positive charge, and the anode which is the negative charge. The electricity generates and flows inside the battery through a wire that connects its two electrodes. In order to control the electrons, positive charges need to travel into another path, shuffling from one electrode to another.
However, the cathode which is made of sulfur gets into a new reaction – the formation of polysulfide molecules. These polysulfide molecules cut the lifespan of a battery because needed sulfur does not go back to the cathode.
Trapping Polysulfide Molecules
Being the number one problem of lithium-ion batteries, experts aspire to address polysulfide molecules, therefore, extending its performance. PNNL’s chemists, headed by Jie Xia, see how the metal organic framework can provide solution to the existing problem.
“The MOF’s highly porous structure is a plus that further holds the polysulfide tight and makes it stay within the cathode,” said PNNL electrochemist Jianming Zheng.
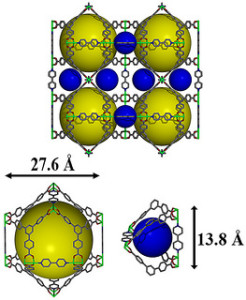
Understanding Metal Organic Framework
Metal Organic Framework has several elements, but chemists from PNNL chose transition metal nickel as its primary element because of its capability to strongly attract sulfur. It is also composed of metal clusters and organic molecules, creating a 3D structure.
PNNL conducted several laboratory tests so as to ensure that MOFs can really trap polysulfide molecules, resulting to extended battery life. According to the tests, the lithium-ion battery maintained 89% of its power even after 100 charge and discharge cycles. Even though it has shown good results, PNNL still aims to improve the MOF so the battery can hold more energy.
“MOFs are probably best known for capturing gases such as carbon dioxide,” Xiao said. “This study opens up lithium-sulfur batteries as a new and promising field for the nanomaterial.”
The innovation of MOFs is funded by the Department of Energy’s Office of Energy Efficiency and Renewable Energy.
With such improvements on the lithium-ion batteries, the performance of electric vehicles could certainly surpass the performance of conventional vehicles. It can certainly result in economic benefits.
Related Article: